Genetic Engineering and Biotechnology is an important topic in Science and Technology that focuses on modifying genetic material and utilizing biological systems for practical applications. It has transformed fields such as medicine, agriculture, industry, and environmental science. Under this topic, we will study the principles, techniques, applications, and significance of genetic engineering and biotechnology in modern life.
Fundamentals of Genetic Engineering and Biotechnology
Biotechnology deals with techniques of using live organisms or enzymes from organisms to produce products and processes useful to humans. Examples: Making curd, bread, wine, vaccines. The two core fields that enabled the birth of modern biotechnology are:
- Genetic Engineering: It includes techniques to alter/change the chemistry of genetic material (DNA and RNA) to introduce these into host organisms and thus change the traits of the host organism.
- Bioprocess Engineering: Bioprocess engineering is the process of keeping the production environment completely clean and free from unwanted microbes so that only the desired microorganism or cell can grow in large amounts and make useful biological products (e.g., antibiotics, vaccines, enzymes).
Biotechnology Areas
| Colour Classification | Field / Area | Important Examples / Uses |
| Blue Biotechnology | Marine & Aquatic Biotechnology | Increase seafood production, control harmful water-borne organisms, development of new drugs. |
| Green Biotechnology | Agricultural Biotechnology | Transgenic/GM crops, improved nutritional quality, higher yield, eco-friendly products. |
| Red Biotechnology | Medical & Health Biotechnology | Production of insulin, antibiotics, vaccines, therapeutic enzymes. |
| White Biotechnology | Industrial Biotechnology | Use of enzymes as industrial catalysts, eco-friendly chemical production. |
Cell organelles and their functions
| Cell organelles | functions |
| Nucleus | DNA storage |
| Mitochondria | Energy production (ATP)Powerhouse of the cell |
| Ribosomes | Protein production |
| Lysosomes | Break down and recycle cellular waste (cell’s digestive system)Protein destructionSuicide bags |
| Vacuole | Store water, nutrients, and the waste materials of the cell. |
Plant Cell vs Animal Cell
| Feature | Plant Cell | Animal Cell |
| Cell Wall | Present (outer cover, made of cellulose) + inner cell membrane | Absent, only cell membrane present |
| Vacuole | Large, single central vacuole (maintains turgor pressure, stores nutrients) | Many small vacuoles, temporary in nature |
| Chloroplasts | Present, site of photosynthesis (contain chlorophyll) | Absent, no photosynthesis |
| Shape | Generally rigid, rectangular shape (due to cell wall) | Generally round, irregular shape |
| Lysosomes | Rarely present (if present, less prominent) | Numerous and prominent |
| Centrioles | Absent (except in lower plant forms) | Present, important for cell division |
| Mode of Nutrition | Autotrophic (photosynthesis possible) | Heterotrophic (depends on intake of food) |
Basics of Genetics
- Genetics: The branch of biology which deals with the study of heredity and variation is called genetics. The term genetics was first coined by Bateson (1905).
- Gene: A gene is a segment of DNA that contains the necessary information to produce a functional product, typically a protein. Genes can vary in size and function, with humans estimated to have about 19,000-25,000 genes.
- Allelomorph or allele: Alternative form of a gene which controls any one character (like colour, height) is called allelomorph or allele. Example: Height of the plant is controlled by a gene which has two alleles T (Tallness) and t (Dwarfism).
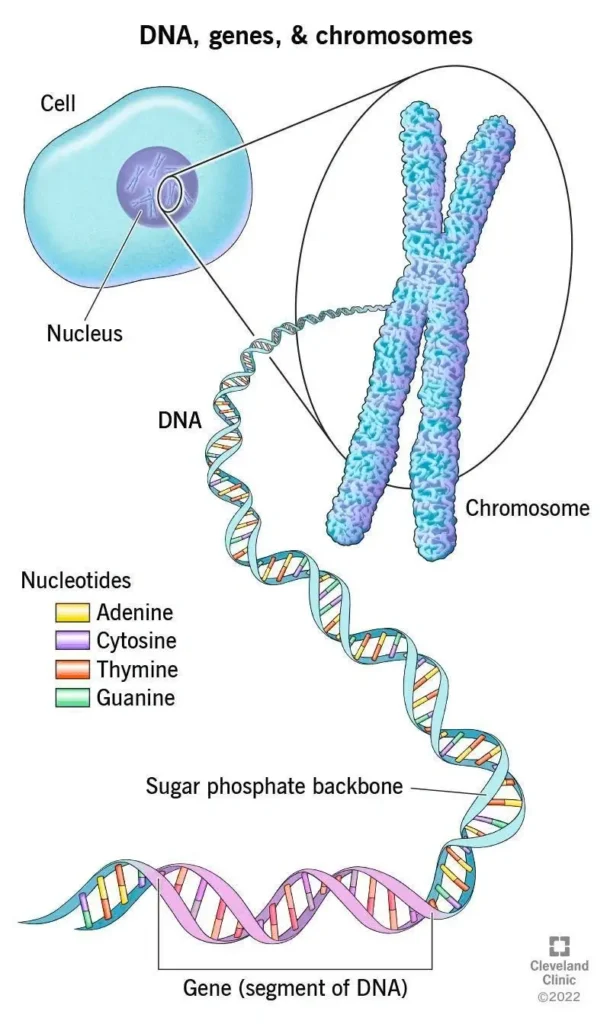
- Chromosome
- In the nucleus of each cell, the DNA molecule is packaged into thread-like structures called chromosomes.
- They carry genetic information in the form of genes, which determine the hereditary traits passed from one generation to the next.
- Humans have 46 chromosomes, organized into 23 pairs.
- Genotype (backend): Pattern of genes in an organism’s DNA responsible for particular traits.
- Phenotype (frontend): Observable physical properties of an organism.
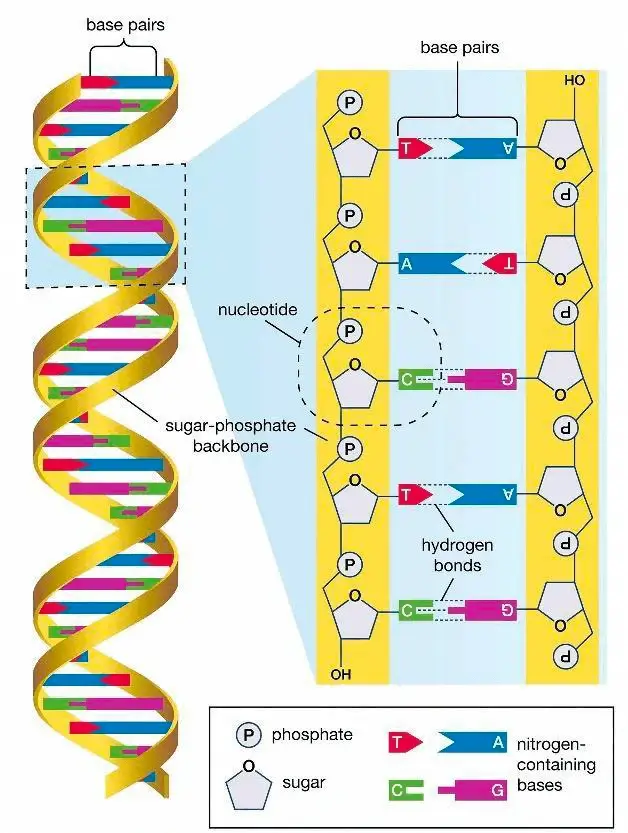
- Nucleic Acids:
- Polymers of nucleotides.
- Each nucleotide is made up of three main components:
- Sugar Molecule: Provides structural support.
- Phosphate Group: Links nucleotides together.
- Nitrogenous Base: Determines the genetic code. Example: Adenine (A), Guanine (G), Cytosine (C), Thymine (T) (Only in DNA) and Uracil (U) (In RNA).
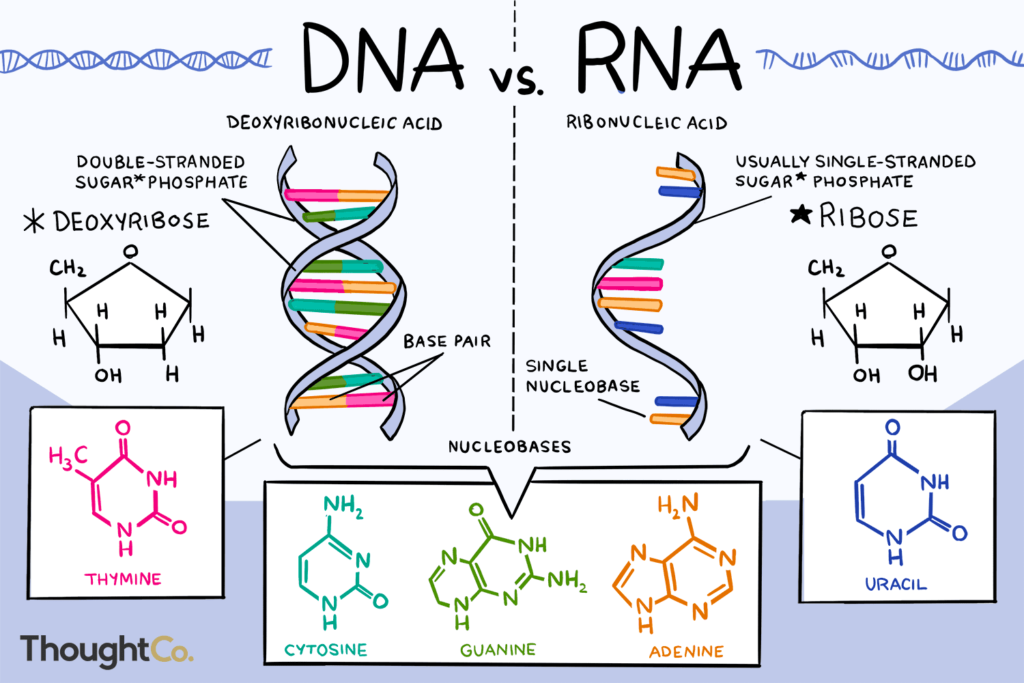
- Nucleic Acids are of two types
- DNA (Deoxy ribonucleic Acid)
- RNA (Ribonucleic Acid).
Key Differences Between DNA and RNA
| Feature | DNA (Deoxyribonucleic Acid) | RNA (Ribonucleic Acid) |
| Sugar | Deoxyribose | Ribose |
| Structure | Double-stranded helix | Single-stranded |
| Nitrogen Bases | Adenine (A),Thymine (T), Guanine (G) and Cytosine (C). | A, U, G, C (Uracil replaces Thymine) |
| Location | Nucleus & mitochondria | Nucleus, cytoplasm, ribosomes |
| Function | Long-term genetic storage | Helps in transfer and expression of information → Protein synthesis (mRNA, tRNA, rRNA) |
| Stability | More stable | Less stable (due to the presence of an extra -OH group), short-lived |
| Replication | Self-replicating | Synthesized from DNA |
Some Important Updates Related to DNA
Mitochondrial DNA (mtDNA)
| Aspect | Details |
| Location | Found in mitochondria (cell cytoplasm), separate from nuclear DNA. |
| Structure | Small, circular, double-stranded DNA (~16,500 base pairs; 37 genes in humans). |
| Genes (37) | 13 proteins (oxidative phosphorylation) + 22 tRNAs + 2 rRNAs (mitochondrial protein synthesis). (mRNA → absent) |
| Inheritance | Maternal inheritance → transmitted almost exclusively from mother. |
| Function | Energy production (ATP), metabolism regulation, signaling, apoptosis (programmed cell death). |
| Differences from Nuclear DNA | – Separate genome, circular (vs linear in nucleus).- Lacks histones → mutates faster than nuclear DNA. – Smaller, limited coding capacity. – Inherited exclusively from the mother (Nuclear DNA → paternal + maternal). |
| Medical Importance | Mutations cause mitochondrial diseases (e.g., Leigh syndrome, MELAS). |
| Other Significance | Used in forensics (maternal lineages, degraded samples), ancestry studies, linked to aging & cancer. |
Dark DNA
- Dark DNA refers to genomic regions that are difficult to detect or interpret using standard sequencing methods, often due to high mutation rates or repetitive sequences. Recent research suggests that dark DNA may play roles in gene regulation, evolution, and species adaptation, though its precise functions remain under investigation.
- Junk DNA refers to non-coding DNA whose function was historically unknown.
Cell-free DNA (cfDNA)
| Aspect | Details |
| Definition | Small fragments of nucleic acids that are released from cells and found outside the cell in body fluids (blood, urine, saliva, cerebrospinal fluid). |
| Source | Released from cells undergoing apoptosis (programmed death) or necrosis (injury/disease-related death). |
| Nature | Degraded, non-functional DNA fragments.Cannot replicate or perform cellular processes. |
| Key Feature | Retains genetic information of the original cell → acts as a “molecular signature.”cfDNA quantity in the blood increases under pathological conditions such as auto-immune diseases, cancer etc. |
| Applications | Prenatal testing (NIPT): Detects fetal chromosomal abnormalities (e.g., Down Syndrome).Oncology (Liquid biopsy): Cancer detection, prognosis, monitoring treatment response.Transplant medicine: Detects donor-derived cfDNA → helps monitor organ rejection.Infectious diseases: Detects pathogen DNA fragments in blood.Used as a biomarker for neurological disorders like Alzheimer’s disease. |
Major Types of RNA
- mRNA (Messenger RNA)
- Copies genetic information from DNA (transcription) and carries it to ribosomes for protein synthesis.
- Transcribed using the enzyme RNA polymerase.
- tRNA (Transfer RNA)
- Brings amino acids to ribosome during translation.
- Reads mRNA in triplets (codons – 3 nucleotide sequences) → each codon codes for one amino acid.
- rRNA (Ribosomal RNA)
- Structural & catalytic component of ribosomes.
- Facilitates peptide bond formation (acts as a ribozyme).
Nobel Prize in Medicine 2024 (Victor Ambros and Gary Ruvkun)
|
For What? |
|
|
About Post-Transcriptional Gene Regulation |
|
|
About microRNA |
|
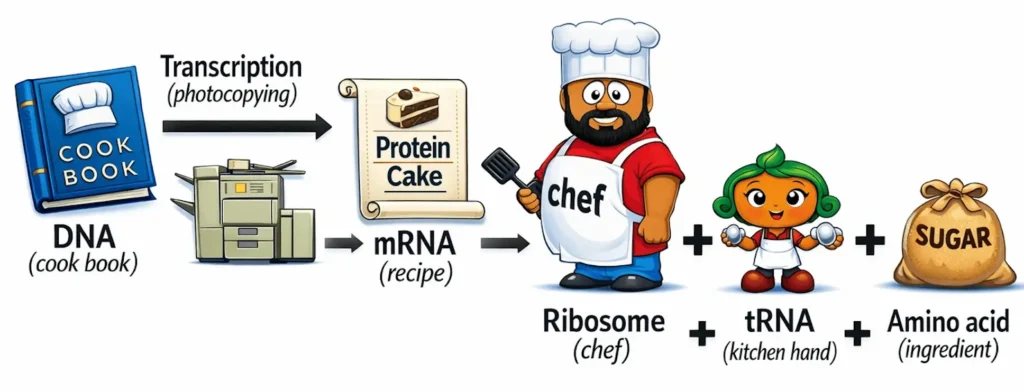
Protein Synthesis (Central Dogma of Molecular Biology)
- Describes the flow of genetic information in cells: DNA → RNA → Protein.
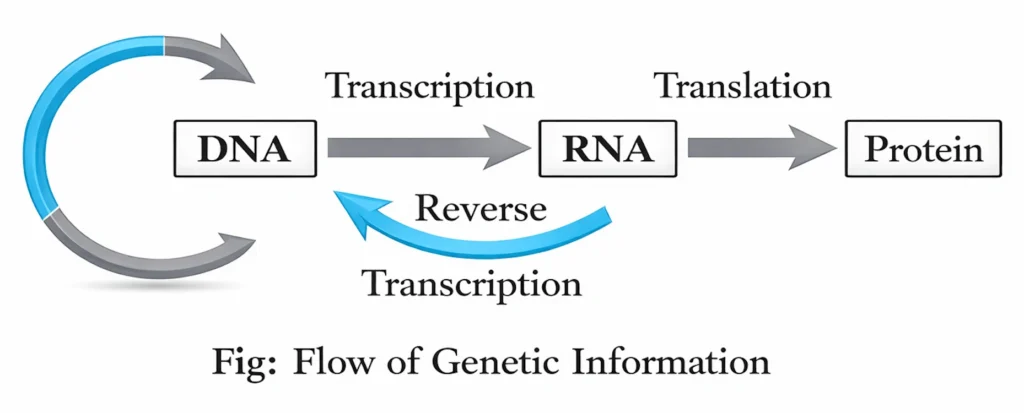
- Gene Expression: The process by which the instructions in DNA are converted into functional proteins. Takes place in two main stages:
- Transcription (Occurs in the nucleus)
- mRNA copies the sequence of a gene from DNA.
- Enzyme: RNA Polymerase.
- Output: mRNA (messenger RNA) leaves nucleus → cytoplasm.
- Translation (Occurs in the cytoplasm at the ribosomes)
- Process: Ribosomes read mRNA sequence.
- Transfer RNA (tRNA) brings amino acids.
- Result: Protein synthesis.
- Transcription (Occurs in the nucleus)
Genome Sequencing
- Genome sequencing is the process of determining the exact order of nucleotides (A, T, C, G) in an organism’s complete set of DNA.
- Genome: Organism’s complete set of DNA.
- Genome includes both coding & non-coding DNA.
- Human genome = ~3 billion base pairs across 23 pairs of chromosomes.
- Whole Genome Sequencing (WGS): A laboratory method that reads the order of all four bases across the entire genome in a single process.
Important Genome Sequencing Initiatives
| Project / Initiative | Description / Objective |
| Human Genome Project (1990-2003) | USA-led projectLed to the decoding of the entire human genome for the first time, giving the ability to read nature’s complete genetic blueprint. |
| IndiGen(2019) | Initiated by CSIR from April 2019 to October 2019.Aim: To sequence whole genomes of 1029 individuals from diverse ethnic groups across India. |
| Genome India Project (2020-2023) | Department of BiotechnologyTo create a comprehensive reference database of genetic variations and subsequent data analysis of 10,000 individuals representing India’s diverse population.Entire dataset will be stored at Indian Biological Data Centre (India’s only data bank for life-sciences – Faridabad, Haryana) and made available as a digital public good or research. |
| One Day One Genome Initiative | Department of Biotechnology and Biotechnology Research and Innovation Council (BIRAC)It will highlight the unique bacterial species found in India, and emphasise their critical roles in environment, agriculture and human health. |
| Telomere to Telomere (T2T) consortium | It sequenced the remaining 8% of human genome. |
| INDIGAU | Genomics of cowIndia’s first high density SNP chip for the conservation of indigenous cattle breed. |
| National Genomic Grid | Aim: To collect cancer cells and tissues to facilitate cancer research in India. Uses genome sequencing of cancer cells to study genomic factors influencing cancer in the Indian population |
| DeepVariant: AI ingenomics | Google’s AI system that converts sequencing data from high-throughput sequencing into an accurate picture of the entire genome. |
| AlphaFold (Nobel Prizein Chemistry 2024) | Google’s AI system that is capable of predicting protein modelling. It is important to understand diseases and corresponding drug development. |
Tmesipteris oblanceolata
|
What is it? |
|
|
Genome Size |
|
|
C-value Paradox |
|
Gene Editing
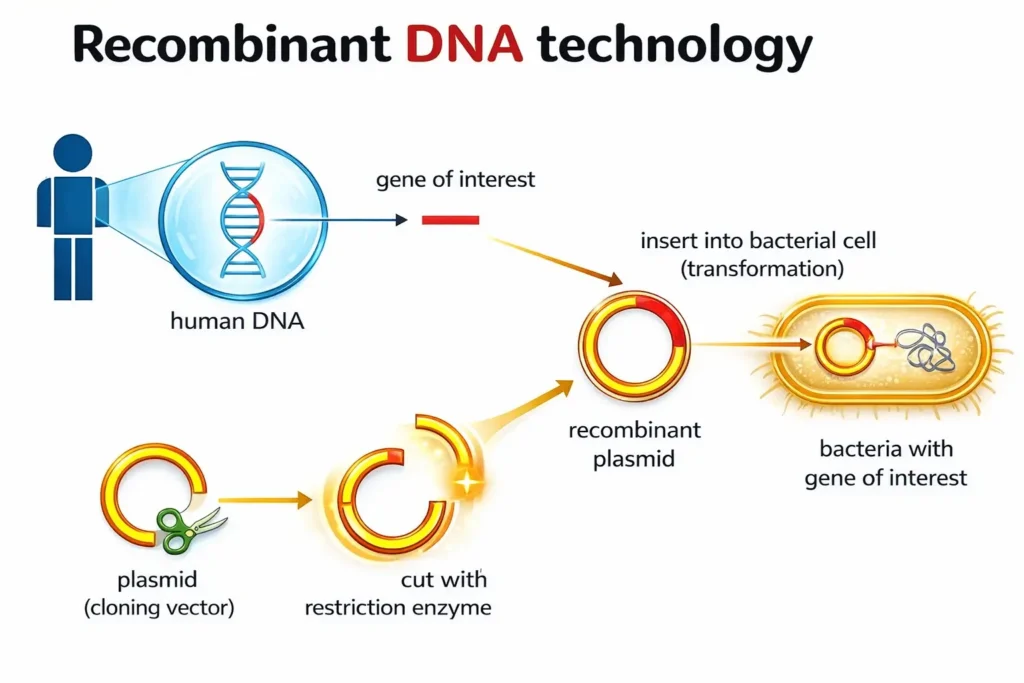
Recombinant DNA Technology
- Direct Manipulation of DNA of an organism to get desired characteristics.
- It involves inserting, deleting, modifying, or replacing DNA in a living organism’s genome.
- Basic principle: Isolating a specific gene or DNA sequence of interest from one organism and inserting it into the genome of another organism. The inserted gene can be from the same species or from a different species.
Steps of Recombinant DNA Technology
- Identification and Isolation of genetic material → extract pure DNA from source organism.
- Cutting DNA at specific sites → use restriction enzymes (e.g. EcoRI) to create fragments.
- Amplification of gene of interest → via PCR (Polymerase Chain Reaction).
- Insertion into vector → ligate DNA fragment into plasmid/viral vector using DNA ligase.
- Transfer into host cell → transformation into bacteria or other cells.
- Selection of recombinant cells → using markers (e.g. antibiotic resistance).
- Expression of foreign gene → production of desired protein.
- Downstream processing → purification and analysis of gene product.
Tools of rDNA technology
|
Name of Tool |
Role in Genetic Engineering |
|
Restriction Enzymes (Restriction Endonucleases) |
|
|
DNA Ligase |
|
|
Vectors |
|
|
Host Organisms |
|
|
Gene Delivery Methods (Transformation Tools) |
|
|
Gel Electrophoresis |
|
|
DNA Polymerase |
|
|
Gene Editing/ Gene Modification Techniques |
|
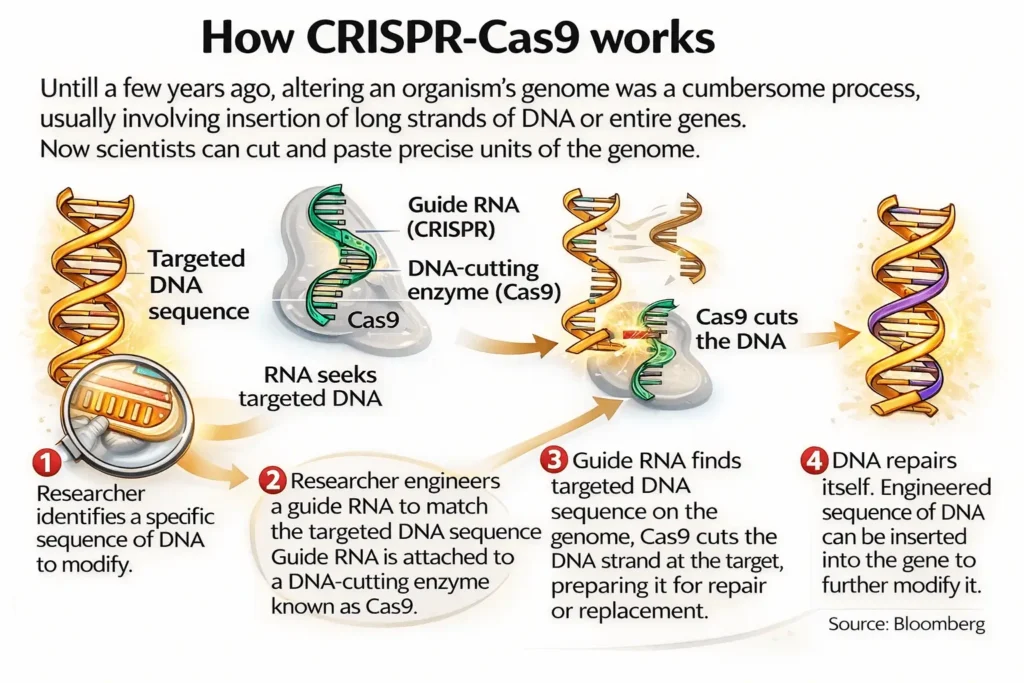
CRISPR-Cas9 Technology
- Clustered Regularly Interspaced Short Palindromic Repeats (CRISPR)
- CRISPR is a powerful tool for editing genomes, allowing researchers to easily alter DNA sequences and modify gene function.
- Cas9: CRISPR-associated protein 9
- Cas9 is an enzyme that acts like a pair of molecular scissors in CRISPR technology.
- Cas9 + guide RNA → precisely targets and cuts DNA at a specific sequence.
- Enables correction of defective genes or insertion of new genes.
- #Recent development: Alternative to Cas9 and Cas12 proteins- ISDra2TnpB (derived from a bacteriam, Deinococcus radiodurans – capable of surviving extreme environmental conditions).
Project to develop CRISPR-based gene-editing therapies to cure sickle cell anemia
- Five-year project started in 2021.
- CSIR is developing the therapies under its Sickle Cell Anaemia Mission.
- This is the first disease to be targeted for CRISPR-based therapy in India.
- CSIR’s Institute of Genomics and Integrative Biology has indigenously developed a CRISPR-based therapeutic solution for sickle cell anaemia, which is now being readied for clinical trials.
Applications of Recombinant DNA Technology
- Medicine and Health Care:
- Production of Therapeutic Proteins:
- Human insulin was the 1st therapeutic protein to be genetically cloned in E-coli.
- Creation of Monoclonal antibodies.
- Vaccines: Hepatitis B vaccine (produced using yeast). Ongoing research for recombinant vaccines against HIV, malaria, and COVID variants.
- Gene therapy:
- Correcting defective genes using rDNA tools (e.g., CRISPR-Cas9) (e.g., Haemophilia).
- The first commercial gene therapy product approved for cancer treatment by China in 2003, was Gendicine.
- Genetic testing (Diagnostics): PCR-based tests for genetic disorders, infectious diseases (HIV and Hepatitis).
- Functional Genomics → Study gene functions.
- Production of Therapeutic Proteins:
- Crop Production and Agriculture
- Crops engineered for drought, salinity, and insect, herbicide, virus resistance; delayed fruit ripening (Flavr Savr tomato).
- Genetically Modified (GM) Crops: Bt cotton (resistant to bollworm), Golden Rice (enriched with Vitamin A).
- Nutritional Improvement: Biofortification using rDNA technology.
- Edible vaccines.
- Biofuels: Bio-butanol → possible replacement for gasoline.
- Crops engineered for drought, salinity, and insect, herbicide, virus resistance; delayed fruit ripening (Flavr Savr tomato).
- Food processing: Fermented foods with added taste, nutrition and shelf life such as cheese, yoghurt, certain probiotics, buttermilk.
- Environmental protection
- Bioremediation: Engineered bacteria degrade oil spills, plastics, and toxic waste.
1. Crop Production and Agriculture
What is Genetic Engineering in Crops?
| Feature | Description |
| Definition | Altering DNA of crops to enhance traits |
| Techniques | Recombinant DNA, CRISPR, gene silencing |
| Traits Modified | Pest resistance, shelf life, nutrition, drought tolerance |
GM Crops: Recent Developments
|
Genetically Modified Crop |
Characteristics / Uses |
|
Bt cotton |
|
|
HTBT Cotton (Herbicide Resistant Bt Cotton) |
|
|
Bt Brinjal |
|
|
GM Rubber |
|
|
GM Soymeal |
|
|
GM bacteria for nitrogen fixation |
|
|
Golden Rice |
|
|
Edible vaccines |
|
|
Protato |
|
DMH 11 (Dhara Mustard Hybrid)
- GEAC (Genetic Engineering Appraisal Committee) has approved its environmental release.
- It is a transgenic hybrid crop developed by researchers at Delhi University by crossing Indian Mustard Varuna (Barnase Line) with East European Early Heera 2 Mutant (Barstar Line).
- Foreign genes come from a soil bacteriam called ‘Bacillus amyloliquefaciens’.
- Note:
- Under normal conditions mustard plants do not cross across varieties as they are self-pollinating in nature.
- As a result we do not have hybrid varieties of mustard due to which plant-breeders are not able to induce desired traits in mustard plants.
- Benefits:
- Increase in yield up to 20-30%.
- Seeks to attain self-sufficiency in edible oil seeds production.
- Issues:
- The transgenic hybrid variety becomes tolerant to a herbicide called glufosinate-ammonium upon genetic modification.
- Herbicides are extensively used in field trials.
- These may harm honey bees (the biggest pollinators of mustard fields).
Flavr Savr Tomato
- Flavr Savr tomato, developed by Calgene Inc., was the first GMO crop approved for human consumption. Commercialized in 1994 in the USA.
- Benefit: Genetic modification suppressed the polygalacturonase enzyme, which softens tomatoes. It allowed vine-ripening (better taste) while maintaining firmness for transport.
- Limitations: Despite improved texture and shelf life, flavor quality was suboptimal. Consumers found taste lacking compared to traditional vine-ripened tomatoes.
India becomes the first country to develop genome-edited rice varieties.
|
Parameter |
Information |
|
Varieties Developed |
|
|
Technology Used |
|
|
|
|
Target States |
|
|
Significance |
|
“Minus 5 and Plus 10” Formula
- Reduce rice cultivation area by 5 million hectares and increase rice production by 10 million tons in the same area.
- Purpose: Free up land for pulses and oilseeds cultivation.
GM Crops Regulators in India
Genetically modified (GM) crops in India are regulated by the Ministry of Environment, Forest and Climate Change (MoEFCC) under the Environment Protection Act of 1986.
- The Department of Biotechnology (DBT) under the Ministry of Science and Technology is the nodal for GM research and setting of safety standards.
- GEAC – Genetic Engineering Appraisal Committee is the regulator under MoEFCC to approve GM Crops in India, allow field trials and approve large scale release in the environment.
- ICAR – Indian Council For Agricultural Research under Ministry of Agriculture for R&D on GM crops.
- Food Safety and Standards Authority of India (FSSAI) – GM foods are also regulated by the FSSAI under the Food Safety and Standards Act of 2006.
Genetic Engineering Appraisal Committee (GEAC)
- GEAC is a statutory body under Environment (Protection) Act, 1986.
- Functions under Ministry of Environment, Forest and Climate Change (MoEF&CC).
- Composition:
- Chaired by MoEF&CC official
- Co-chaired by DBT representative
- Members: Includes experts from ICAR, ICMR, CCMB etc.
- Responsible for approving:
- Large-scale use of GM organisms in research/industry.
- Environmental release of GM crops (e.g. Bt cotton).
- Clearance from GEAC is mandatory before commercial use of GM products (Ex. Bt Cotton).



Important Hybrid Crop Varieties
| Crop Variety | Key Feature / Significance |
| Pusa Vivek QPM 9 | First high vitamin-A maize hybrid |
| DMRH 1308 | A high yielding maize hybrid for wider adaptability |
| Girnar 5 | Rich in oleic acid hybrid groundnut |
| Virat (IPM 205-7) | The world’s first extra early synchronous variety of mungbean |
| IPL 220 | Biofortified Lentil Variety |
| JRO 524 | Jute variety exported to Bangladesh |
| PSL-17 | A variety of Pulse (Lentil). |
Food Fortification
- Deliberately increasing the content of essential micronutrients in food during processing to improve the nutritional quality to provide public health benefit with minimal risk to health.
- In India,
- Wheat flour & Rice → Iron, Vitamin B12, Folic Acid
- Milk & Edible Oil → Vitamins A & D
- Double Fortified Salt → Iodine + Iron.
Fortified Rice
- Technologies: Coating, dusting, and extrusion.
- The Cabinet Committee on Economic Affairs accorded its approval for supply of fortified rice throughout the Targeted Public Distribution System (TPDS).
- 10 g of FRK must be blended with 1 kg of regular rice.
- Fortified rice will be packed in jute bags with the logo (‘+F’) and the line “Fortified with Iron, Folic Acid, and Vitamin B12”.
Biofortification
- Breeding crops to increase their nutritional value.
- This can be done either through conventional selective breeding, or through genetic engineering.
- Example:
- Golden Rice (biofortified with Vitamin A, not yet widely adopted in India).
- Iron-rich pearl millet (Dhanashakti).
- Zinc-rich rice (DRR Dhan 45) and wheat varieties released under ICAR programmes.
- Protein-rich maize.
- Vitamin A rich sweet potato.
2. Medicine and Health Care
A. Gene Therapy
- Gene therapy is a rapidly advancing field in biotechnology that aims to treat genetic diseases by modifying the expression of disease-causing genes.
- It involves introducing healthy genes to replace or supplement faulty ones and correcting genetic abnormalities.
- Unlike traditional drugs, gene therapy targets the root genetic causes inside cells.
- Types of Gene Therapy
|
Somatic cell gene therapy |
Germline gene therapy |
|
|
- Gene therapies can have an autologous or allogeneic source of cells:
- Autologous: The patient’s cells are used as a source of cells.
- Allogeneic: Cells from a donor are used as a source of cells.
- Gene therapy can be used to treat monogenic disorders caused by a mutation in a single gene, such as SCID, haemophilia, and muscular dystrophy.
- The Department of Biotechnology has successfully executed a first-in-human gene therapy using lentiviral vectors (viral vectors used to transfer genetic material into cells for gene therapy) to treat Haemophilia-A.
- First human clinical trial of gene therapy to treat Haemophilia A (FVIII deficiency) at Christian Medical College (CMC) Vellore (Tamil Nadu).
USFDA approved 2 gene therapies for sickle cell disease: Casgevy, Lyfgenia.
- Casgevy is based on CRISPR which uses molecular “scissors” to trim faulty parts of genes that can be disabled or replaced with new strands of normal DNA.
- Lyfgenia uses a more conventional form of gene therapy that uses a virus to transfer a gene into cells.
Luxturna
- First FDA-approved gene therapy for an inherited retinal disease (IRD).
- Uses Adeno-Associated Virus (AAV2) vector to deliver a functional copy of the RPE65 gene.
Enzyme Replacement Therapy
- U.S. Food and Drug Administration (USFDA) recently gave nod to world’s first enzyme replacement therapy (ERT).
- Adzynma – The first genetically engineered protein product for ERT.
- For treating congenital thrombotic thrombocytopenic purpura (cTTP), a rare blood clotting disorder.
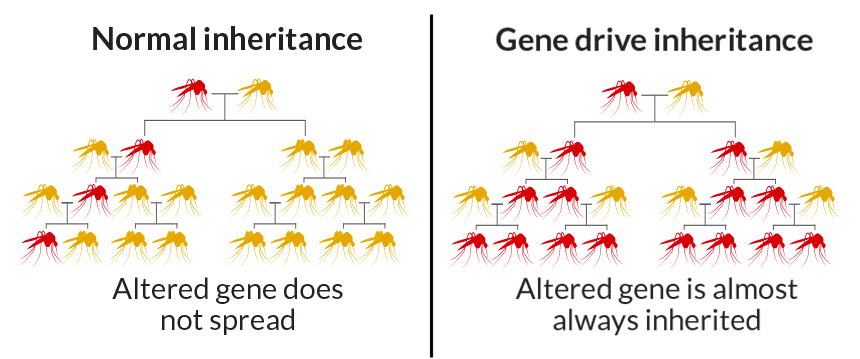
B. Gene Drive Technology
- Genetic engineering technique that modifies genes so that they don’t follow the typical rules of heredity.
- It increases the likelihood that a particular suite of genes will be passed onto the next generation, allowing the genes to rapidly spread through a population and override natural selection.
- Normal Inheritance vs. Gene Drive
- Normal: 50% chance of a gene being inherited from either parent.
- Gene Drive: Specific gene copies itself onto both chromosomes, increasing inheritance likelihood (~100%).
- Applications: Agricultural pest control, disease vector control.

- Application Example
- Target: Female mosquitoes (malaria vector).
- Gene: ‘doublesex’ gene edited via CRISPR → Disrupts normal female development.
- Outcome: Inherited trait leads to sterile or male-like mosquitoes incapable of biting or reproducing. → Complete elimination of female mosquitoes within about 8 generations → Reduces malaria transmission.
Wolbachia Method
- A biological control strategy to reduce mosquito-borne diseases like Dengue, Zika, and Chikungunya.
- Uses Wolbachia, a naturally occurring bacteria found in many insect species, but not in Aedes mosquitoes (disease carriers).
- Infected Males + Wild Females → Eggs do not hatch (reduces mosquito numbers).
- Infected Females + Any Male → Offspring inherit Wolbachia, spreading the trait.
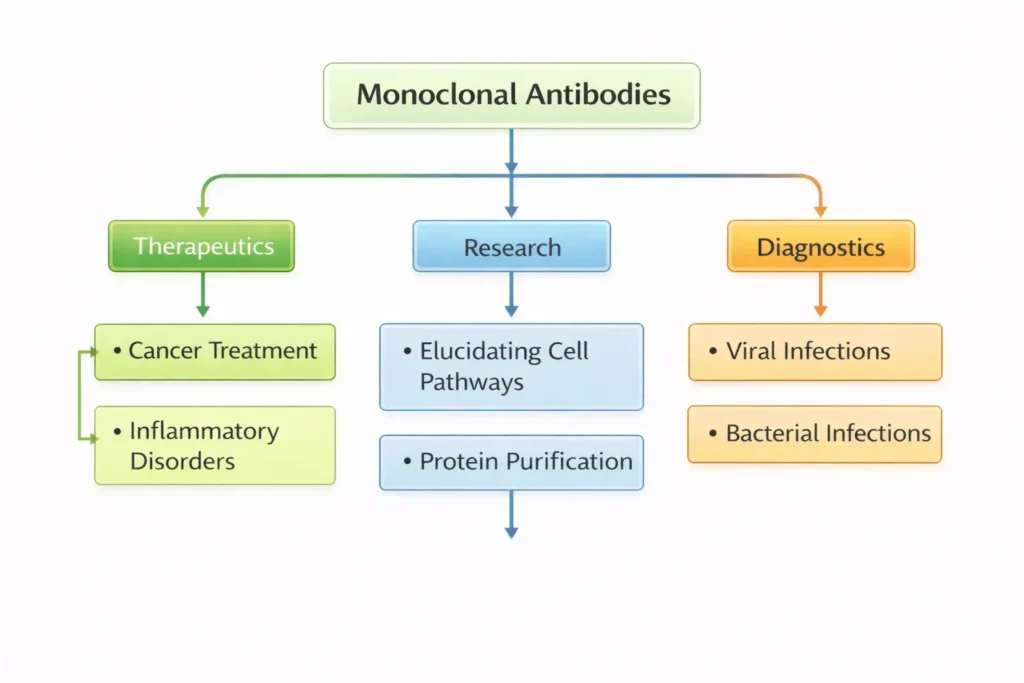
C. Monoclonal Antibodies
- Laboratory-made (man-made) proteins to serve as substitute antibodies and mimic their behaviour to protect against diseases and foreign substances.
- They are engineered to specifically bind to a particularantigen (disease-causing molecule) and neutralise it.
Difference between Antibody & Monoclonal Antibody
| Feature | Antibody | Monoclonal Antibody |
| Source | Naturally produced by the immune system (B-cells) during infection. | Produced in laboratories using biotechnology (hybridoma or recombinant DNA technology). |
| Specificity | Polyclonal – targets multiple antigens | Monospecific – targets a single antigen |
| Example | Antibodies produced during any infection | mAbs used in Covid-19, cancer therapy |
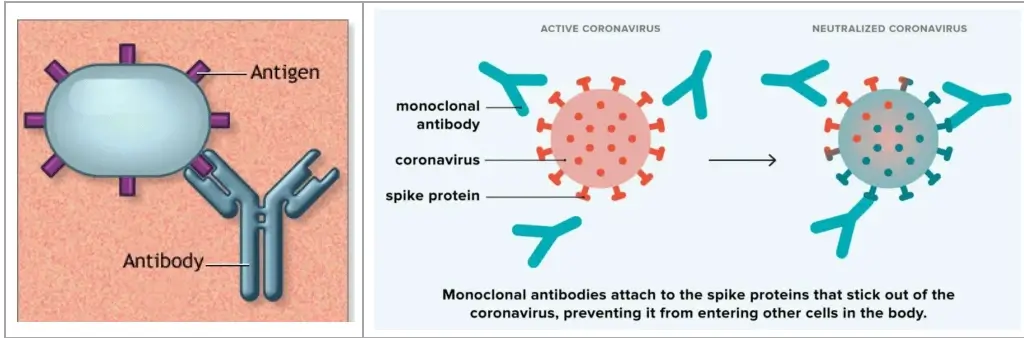
Applications of Monoclonal antibodies
- Nipah virus: Recently, India reached out to Australia to procure monoclonal antibody doses to combat the Nipah virus outbreak in Kerala.
- Covid-19: Monoclonal antibodies during COVID-19 pandemic were engineered to bind to the spike protein of SARSCoV-2 virus. → Prevent virus entry into cells → Reduce severity of disease.
- Cancer: They can target specific cancer cells. → Block growth signals or mark for immune destruction.
D. Vaccines
Types of Vaccination
|
Type of Vaccine |
Characteristics / Examples |
|
Inactivated |
|
|
Live-attenuated |
|
|
mRNA |
|
|
Subunit |
|
|
Toxoid |
|
|
Viral vector |
|
Vaccine Technology Advancements in India
| Vaccine Name | Key Features |
| ZyCoV-D (2021) | World’s first and India’s indigenously developed DNA based vaccine for COVID-19 developed by Zydus Cadila. |
| CORBEVAX (2022) | India’s first protein subunit COVID-19 vaccine, by Biological E. |
| iNCOVACC (2023) | Needle-free intranasal COVID-19 vaccine, developed by Bharat Biotech. |
| GEMCOVAC-19 (2023) | India’s first mRNA vaccine for COVID-19, developed by Gennova Biopharma. |
| Other Important Vaccines | |
| CERVAVAC (2023) | India’s first indigenous HPV vaccine for cervical cancer, by Serum Institute.Type: Protein subunit recombinant vaccine.Quadrivalent: Against HPV types 6, 11, 16, and 18.CERVAVAC is based on VLP (Virus-Like Particles). |
Mosquirix (RTS,S/AS01)
- World’s first malaria vaccine.
- Type: Recombinant protein-based vaccine.
- Target: Plasmodium falciparum – the deadliest malaria parasite.
- Developer: GlaxoSmithKline (GSK), with support from PATH and WHO.
- Cameroon became the first country to launch the vaccine as part of their routine immunisation programme.
R21/Matrix-M Vaccine
- Malaria vaccine for children.
- Type: Recombinant protein-based vaccine with Matrix-M adjuvant.
- Developer: University of Oxford + Serum Institute of India (SII).
TB Vaccine (M72/AS01E)
- In Phase III trials, backed by Gates Foundation.
Some Terms Related to Vaccines
- Inverse Vaccine
- In autoimmune diseases, the immune system attacks healthy tissue e.g., psoriatic disease.
- Inverse vaccine stops the immune system from attacking good, healthy cells.
- Self-amplifying RNA (saRNA): CEPI funded optimization for faster, lower-dose vaccines.
- Virus-Like Particles (VLPs):
- Safe and effective mimicry of pathogens.
- Used for HPV, COVID boosters, and emerging pathogens.
- CERVAVAC is based on VLP (Virus-Like Particles).
- T-cell Based Vaccines: Focused on durable immunity beyond antibodies.
- Combination Vaccines: COVID + Flu + RSV in single dose formulations.
3. Environmental protection
Bioleaching
- Bioleaching = Extraction of metals from ores using microorganisms (bacteria, fungi).
- It is an eco-friendly alternative to conventional smelting and chemical leaching.
Bioremediation
- It is a process that uses living organisms (microorganisms, fungi, plants) to degrade, remove, or neutralize pollutants in soil, water, and air.
- Pilot projects in Rajasthan for arsenic removal from groundwater using bioremediation.
- There are two main types of bioremediation:
1. In situ Bioremediation:
- This involves treating the contaminated material at the site without removing it.
- Examples:
- Oil spills → Bacteria (Alcanivorax borkumensis) degrade hydrocarbons in seawater.
- Groundwater contamination → Microbes break down chlorinated solvents.
2. Ex situ Bioremediation
- This method involves removing the contaminated material (soil, water, etc.) from the polluted site and treating it elsewhere, usually in a controlled environment like a bioreactor or compost pile, where the contaminants are broken down more efficiently.
- Examples:
- Bioreactors: Soil or water is pumped through a reactor containing microorganisms that degrade pollutants.
- Composting: Contaminated soil is mixed with organic material and microorganisms, speeding up the breakdown of contaminants.
Mechanisms of Bioremediation
- Bioaugmentation: Addition of specific microbes to degrade pollutants.
- Biostimulation: Adding nutrients to stimulate indigenous microbial growth.
- Phytoremediation: Use of plants to absorb/degrade pollutants.
- Mycoremediation: Use of fungi to degrade pollutants.
