Basics of everyday science is an important topic in Science and Technology that explains the scientific principles behind day-to-day phenomena such as motion, heat, light, electricity, and common chemical reactions. It helps build scientific understanding, logical thinking, and the practical application of science in daily life.
Science is a way of thinking, observing and doing things to understand the world we live in and to uncover the secrets of the universe.
Physics in Everyday Life
UNITS AND MEASUREMENT
- Unit : A unit is a fixed and internationally accepted standard used for measurement.
- Fundamental (Base) unit – These units do not depend on other units.
- Fundamental quantity – unit and symbol
- Length – metre (m)
- Mass– kilogram (kg)
- Time – second (s)
- Electric current – ampere (A)
- Temperature – kelvin (K)
- Amount of substance – mole (mol)
- Luminous intensity – candela (cd)
- Besides the seven base units, there are two more units
- The unit for plane angle is radian with the symbol rad and
- The unit for the solid angle is steradian with the symbol sr. Both these are dimensionless quantities.
- Derived Quantities
- Quantities that can be expressed using Fundamental (base) quantities
- Example – Area (m²), Speed – m/s
THE INTERNATIONAL SYSTEM OF UNITS
- In earlier times, different countries used different systems of measurement.
- The most commonly used systems were:
| System | Length | Mass | Time |
| CGS | centimetre (cm) | gram (g) | second (s) |
| FPS | foot (ft) | pound (lb) | second (s) |
| MKS | metre (m) | kilogram (kg) | second (s) |
- SI System (International System of Units) – internationally accepted system of units.
- It was developed by the International Bureau of Weights and Measures (BIPM).
- The latest revision was done in November 2018.
Some important physical quantities and their units
| Quantity | Units |
| Pressure | Pascal (Pa), Atmosphere (atm), Bar, Torr (mmHg) |
| Loudness of sound | Decibel, bel, phon |
| Energy / Work / Heat | Joule (J), Erg, Calorie, Electronvolt (eV), Kilowatt-hour (kWh) |
| Force | Newton (N), Dyne, Kilogram-force (kgf) |
| Power | Watt (W), Horsepower (hp), erg/sec |
| Temperature | Celsius (°C), Kelvin (K), Fahrenheit (°F) |
| Length / Distance | Meter (m), Kilometer, Mile, Foot, Inch, Nautical mile, Light year, AU, Parsec |
| Mass | Kilogram (kg), Gram, Tonne, Pound (lb), Ounce |
| Light / Illumination | Lumen (lm), Candela (cd), Lux |
| Intensive Property | Extensive property |
| The properties of matter that do not depend on the size or quantity of matter in any way. | An extensive property of a system depends on the system size or the amount of matter in the system. |
| Example– Temperatures, density, boiling point, melting point, colour, ductility, conductivity, pressure, lustre, freezing point, odour, refractive index, specific heat capacity, viscosity etc. | Example – length, mass, volume, weight, enthalpy, entropy, Gibb’s energy, internal energy. |
Motion and Laws of Motion
MOTION IN A STRAIGHT LINE
- Motion – An object is in motion if its position changes with time. Motion along a straight line is called rectilinear motion.
- Distance – Total length of the path travelled by an object. It is a scalar quantity (only magnitude). It is always positive or zero.
- Displacement – Shortest straight-line change in position from start to end. It is a vector quantity (magnitude + direction). It can be positive, negative, or zero. Depends only on initial and final position.
- Distance ≥ Displacement always.
- Speed – Rate of change of distance with time. It is a scalar quantity (only magnitude). It is always positive or zero. Tells how fast an object moves.
- Velocity – Rate of change of displacement with time. It is a vector quantity (magnitude + direction). It can be positive, negative, or zero. Tells how fast and in which direction an object moves.
- Acceleration – rate of change of velocity. It can be due to: Change in speed, Change in direction, or Both. It can be positive, negative, or zero.
- Uniform Acceleration – Acceleration remains constant with time. Many motions like free fall are examples of uniform acceleration.
Three Equations of Motion (For motion with constant acceleration)
- v = u + at (Final velocity depends on initial velocity, acceleration, and time)
- S = ut + ½ at² (Displacement depends on initial velocity, time, and acceleration.)
- v² = u² + 2as (Relation between velocity and displacement (time not involved).
Free Fall
- When an object falls only under the effect of gravity and no other force (like air resistance) acts on it, the motion is called free fall.
- Only gravitational force acts on the body. Mass of the object does not affect free fall. Speed increases continuously while falling.
Weightlessness
- When the apparent weight of a body becomes zero, the condition is called weightlessness.
- Note: The actual weight does not become zero, but it cannot be felt.
- During Free Fall – When a person falls freely, the supporting force becomes zero → weightlessness is felt.
- Eg. – Astronauts feel weightless because the satellite and the astronaut both are in continuous free fall around the Earth.
- If a lift falls freely, a person inside feels weightless.
Force and Laws of motions
- Force – A force is an external agency (push or pull) that can: Change the state of rest, Change speed, Change direction, Cause deformation
- Force can act: With contact (friction, tension, normal), Without contact (gravitational, magnetic)
Law of Inertia (Galileo) –
- On a frictionless surface, a moving object will not stop on its own. Rest and straight line motion are mechanically equivalent physical states as no net force is required to maintain either. This understanding led to Newton’s First Law.
Newton’s first law of motion :
- Every body maintains its initial state of rest or motion with uniform speed on a straight line unless an external force acts on it.
- The first law is also called the law of Galileo or law of inertia.
- Inertia: Inertia is the property of a body by virtue of which the body opposes change in its initial state or motion with uniform speed on a straight line. Inertia is of two types 1. Inertia of rest 2. Inertia of motion
- Examples: – When a car or train starts suddenly, the passengers bends backward.
- When a running horse stops suddenly, the rider bends forward.
- When a coat/blanket is beaten by a stick, the dust particles are removed.
Second law of motion:
- The rate of change in momentum of a body is directly proportional to the applied force on the body and takes place in the direction of force. F=ma.
- Example – 1. Rocket Launch, 2. Karate Player Breaking Slab of Bricks , 3. Professional athletes move their hands back once they catch the ball as it provides the ball more time to lose its speed
Third Law of Motion:
- For every action, there is an equal and opposite reaction, but this pair of forces does not require a collision and acts on two different bodies.
- Examples -1. Recoil of a gun, 2.Motion of rocket, 3. Swimming, 4. While drawing water from the well, if the string breaks up the man drawing water falls back, 5. It is difficult to fix a nail on a freely suspended wooden frame.
Principle of conservation of linear momentum:
- If no external force acts on a system of bodies, the total linear momentum of the system of bodies remains constant.
- As a consequence, the total momentum of bodies before and after collision remains the same.
- Rocket works on the principle of conservation of linear momentum.
Impulse
- When a very large force acts on a body for a very small Duration, then force is called impulsive force. Impulse is defined as the product of force and time.
- Impulse = force × time = change in momentum.
- It is a vector quantity and its direction is the direction of force. Its SI unit is newton second (Ns).
Friction and Types of Forces
Friction
- The force which opposes the relative motion between two bodies in contact is called friction. Friction depends upon nature of contact surface. Due to roughness, the friction increases.
- Friction is of two types:
- Static friction – The force of friction which is introduced when there is no relative motion. Static friction is a self adjusting force. Its value becomes maximum when the applied force on its body is such that the body is about to slide. Maximum value of static friction is called limiting friction.
- Kinetic friction – The friction which acts when a body slides over the surface of other body. Kinetic friction is less than the limiting friction. Kinetic friction is independent of relative speed between the bodies provided the speed is less.
- Rolling friction – This kind of friction acts when a body rolls over the surface of other body. Rolling friction is much smaller than the kinetic friction.
Types of forces
| Conservative force | Non-conservative force |
| A force is said to be conservative if the work done by or against force is dependent only on the initial and the final position of the object and not on the path followed by the object.Work done by the conservative force in a closed path is zero.Example: Gravitational force, Magnetic Force, Electrostatic force, Elastic force, etc. | A force is said to be non-conservative if the work done by or against the force is dependent on the path followed by the object.Work done by the non-conservative force in a closed path is not zero.Example: Friction force, Viscous force, etc. |
- Contact forces – Muscular forces, Frictional force
- Non contact force – Magnetic force, Electrostatic force, Gravitational force
Centripetal Force
- When a body travels along a circular path, its velocity changes continuously. Naturally an external force always acts on the body towards the centre of the path.
- The external force required to maintain the circular motion of the body is called centripetal force.
- F=mv^2/R
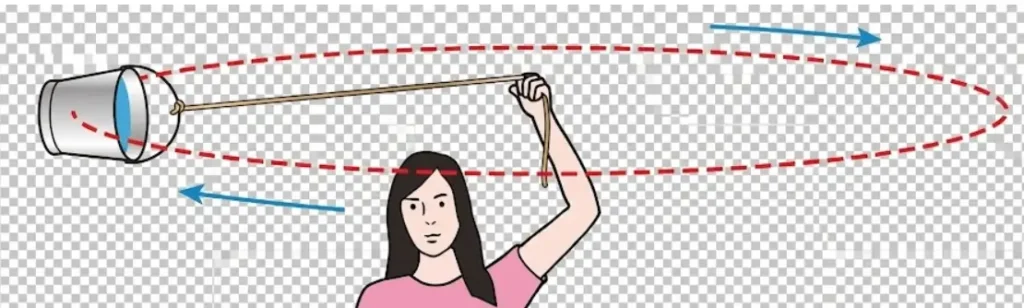
- If a stone attached to a string is whirled in a circular path, the required centripetal force is supplied by the tension in the string.
- For the motion of planets around the sun, the required centripetal force is supplied by the gravitational attraction of sun
- For the motion of electrons around the nucleus in an atom, the required centripetal force is supplied by the electrostatic attraction produced by the proton in the nucleus.
- A cyclist negotiating a curved path bends towards the centre of the path so that the horizontal component of normal reaction on the cyclist by the ground should supply the required centripetal force. Due to this reason the outer edge of a curved road is raised. This is called banking of roads.
Centrifugal Force
- In applying the Newton’s laws of motion, we have to consider some forces which can not be assigned to any object in the surrounding. These forces are called pseudo force or inertial force.
- Centrifugal force is such a pseudo force. It is equal and opposite to centripetal force.
- Cream separator, centrifugal drier work on the principle of centrifugal force.
- Centrifugal force should not be confused as the reaction to centripetal force because forces of action and reaction act on different bodies.
Work, Energy and Power
- Work – If a body gets displaced when a force acts on it, work is said to be done. Work is measured by the product of force and displacement of the body along the direction of force.Work is a scalar quantity and its SI unit is joule.
- W=FScosθ (where θ = angle between force and displacement.)
- In the case of uniform circular motion, work done by the centripetal force is zero.
- Energy – Capacity of doing work by a body is called its energy. Energy is a scalar quantity and its SI unit is joule. Energy developed in a body due to work done on it is called mechanical energy.
- Mechanical energy is of two types:
- Potential Energy – The capacity of doing work developed in a body due to its position or configuration is called its potential energy. Examples: Energy of stretched or compressed spring, Energy of water collected at a height, Stretched bow. Potential Energy of a body in the gravitational field of earth is PE=mgh.
- Kinetic Energy – Energy possessed by a body due to its motion is called kinetic energy of the body. A flying aeroplane has both potential as well as kinetic energy.
Principle of Conservation of Energy
- Energy can neither be created nor can be destroyed. Only energy can be transformed from one form to another form. Whenever energy is utilized in one form, equal amount of energy is produced in other form. Hence total energy of the universe always remains the same. This is called the principle of conservation of energy.
- When a ball is thrown vertically upward its potential energy increases, kinetic energy decreases but the total energy remains constant.
| Equipment | Energy Transformed |
| Dynamo | Mechanical energy into electrical energy |
| Tube light | Electrical energy into light energy |
| Battery | Chemical energy into electrical energy. |
| Electric Bulb | Electrical energy into light and heat energy |
| Solar Cell | Solar energy into electrical energy |
| Loud Speaker | Electrical energy into sound energy |
| Candle | Chemical energy into light and heat energy |
| Electric Motor | Electrical energy into mechanical energy |
| Microphone | Sound energy into electrical energy. |
| Sitar | Mechanical energy into sound energy |
| Photocell | Light energy into electrical energy. |
| Rectifier | Convert alternating current into direct current |
| Inverter | Converts direct current into alternating current |
- Power – Rate of doing work is called power. SI unit of power is watt, named as a respect to the scientist James Watt.
- 1 watt = joule/sec. Horse power is a practical unit of power. 1 H.P. = 746 watt. 1 kilowatt hour (kWh) = 3.6 × 10^6 joule
Gravitation and Space Physics
Gravitation
- Every body attracts other body by a force called force of gravitation.
- Newton’s law of Gravitation – The force of gravitational attraction between two point bodies is directly proportional to the product of their masses and inversely proportional to the square of the distance between them.
- Consider two point bodies of masses 𝑚 1 and 𝑚 2 placed at a distance r. The force of gravitational attraction between them,
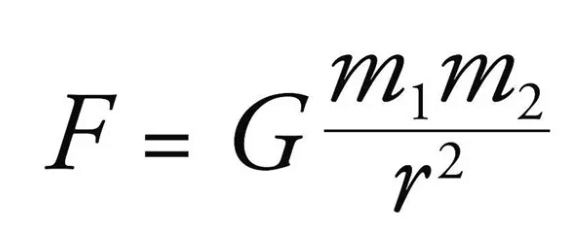
- Gravity – The gravitational force of earth is called gravity. i.e. gravity is the force by which earth pulls a body towards its centre.
- The acceleration produced in a body due to force of gravity is called acceleration due to gravity (denoted as g) and its value is 9.8 m/s².
- Acceleration due to gravity is independent of shape, size and mass of the body. The value of g on moon is less than that on earth. Hence on moon, a man can jump higher than earth.
Variation in g (weight = mg)
- Value of g decreases with height or depth from earth’s surface.
- g is maximum at poles.
- g is minimum at equator.
- g decreases due to rotation of earth.
- g decreases if angular speed of earth increases and increases if angular speed of earth decreases.
- If angular speed of earth becomes 17 times its present value, a body on the equator becomes weightless.
Weight of a body in a lift
- If lift is stationary or moving with uniform speed (either upward or downward), the apparent weight of a body is equal to its true weight.
- If lift is going up with acceleration, the apparent weight of a body is more than the true weight.
- If lift is going down with acceleration, the apparent weight of a body is less than the true weight.
- If the cord of the lift is broken, it falls freely. In this situation the weight of a body in the lift becomes zero. This is the situation of weight-lessness.
- While going down, if the acceleration of lift is more than acceleration due to gravity, a body inside the lift moves upwards relative to the lift and comes in contact with the ceiling of lift.
Kepler’s Laws of planetary motion
- First law – All planets move around the sun in elliptical orbits, with the sun being at rest at one focus of the orbit.
- Second law – The position vector of the planet with sun at the origin sweeps out equal area in equal time i.e. the areal velocity of planet around the sun always remains constant.
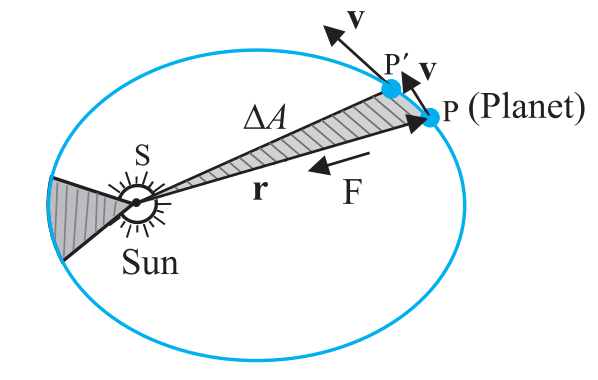
- As a consequence of this law is that the speed of planet increases when the planet is closer to the sun and decreases when the planet is far away from sun. Speed of a planet is maximum when it is at perigee and minimum when it is at apogee.
- Third law – The square of the period of revolution of a planet around the sun is directly proportional to the cube of mean distance of planet from the sun. Clearly distant planets have larger period of revolution. The time period of nearest planet Mercury is 88 days where as time period of farthest planet Pluto is 247.7 years.
Satellite – Satellites are natural or artificial bodies revolving around a planet under its gravitational attraction. Moon is a natural satellite while INSAT-IB is an artificial satellite of earth.
Orbital speed of a satellite
- Orbital speed of a satellite is independent of its mass. Hence satellites of different masses revolving in the orbit of same radius have same orbital speed.
- Orbital speed of a satellite depends upon the radius of orbit (height of satellite from the surface of earth). Greater the radius of orbit, lesser will be the orbital speed.
- The orbital speed of a satellite revolving near the surface of earth is 7.92 km/sec.
Escape velocity
- Escape velocity is that minimum velocity with which a body should be projected from the surface of earth so as it goes out of gravitational field of earth and never return to earth.
- Escape velocity is independent of the mass, shape and size of the body and its direction of projection. Escape velocity is also called second cosmic velocity. For earth, escape velocity = 11.2 km/s.
- For moon, escape velocity = 2.4 km/s.
- Escape velocity is √2 times the orbital velocity. Therefore if the orbital velocity of a satellite is increased to √2 times (increased by 41%), the satellite will leave the orbit and escape.
Fluid Mechanics and Properties of Matter
Pressure
- Pressure is defined as force acting normally on unit area of the surface. SI unit of pressure is N/m² also called pascal (Pa). Pressure is a scalar quantity.
- Application
- The bags and suitcases are provided with broad handles so that small pressure is exerted on the hand while carrying them.
- Railway tracks are laid on large sized wooden sleepers so that thrust due to weight of train is spread over a large areas.
- Pins are made to have pointed ends. This explains why needle is used to brust a gas filled balloon because needle exerts more pressure.
Atmospheric Pressure –
- Pressure exerted by the weight of the air on a unit area of the earth’s surface. Pressure which is exerted by a mercury column of 76 cm length at 0°C at 45° latitude at the sea-level. Weight of 76 cm column of mercury of cross-sectional area 1 cm². Generally it is measured in bar.
- 1 bar = 10⁵ N/m². Atmospheric pressure 1 atm = 1.01 bar = 1.01 × 10⁵ N/m² = 760 torr.
- Atmospheric pressure decreases with altitude (height from earth’s surface).
- This is why – It is difficult to cook on the mountain. The fountain pen of a passenger leaks in aeroplane at height.
- Atmospheric pressure is measured by barometer. With the help of barometer, weather forecast can be made.
- Sudden fall in barometric reading is the indication of storm.
- Slow fall in barometric reading is the indication of rain.
- Slow rise in the barometric reading is the indication of clear weather.
Pressure in liquid
- Force exerted on unit area of wall or base of the container by the molecules of liquid is the pressure of liquid.
Pascal law for pressure of liquid
- If gravitational attraction is negligible, in equilibrium condition, pressure is same at all points in a liquid.
- If an external pressure is applied on an enclosed fluid, it is transmitted undiminished to every direction.
- Hydraulic lift, hydraulic press, Hydraulic brake work on Pascal law.
Floatation
Buoyant Force
- When a body is immersed partly or wholly in a liquid, a force acts on the body by the liquid in the upward direction. This force is called Buoyant force or force of buoyancy or upthrust. It is equal to the weight of liquid displaced by the body and acts at the centre of gravity of displaced liquid. Its study was first made by Archimedes.Archimedes Principle
- When a body is immersed partly or wholly in a liquid, there is an apparent loss in the weight of the body which is equal to the weight of liquid displaced by the body.
Law of Floatation
- A body floats in a liquid if: Density of material of body is less than or equal to the density of liquid.
- If density of material of body is equal to density of liquid, the body floats fully submerged in liquid in neutral equilibrium.
- When body floats in neutral equilibrium, the weight of the body is equal to the weight of displaced liquid.
- The centre of gravity of the body and centre of gravity of the displaced liquid should be in one vertical line.
- When a solid body is dropped from the boat (floating in water), the level of water will
- Fall if density of solid > density of water and body will sink.
- For example when the iron nails are removed from a boat and sink in water. The water level falls.
- If the removed body is less dense than water and floats, the water level remains unchanged.
- When a piece of ice, floating in a liquid melts completely, the level of liquid + water:
- remain unchanged if density of liquid = density of water
- falls if density of liquid < density of water
- rises if density of liquid > density of water
- A piece of solid embedded with an ice block floating in water. If ice melts completely, the level of water
- Falls if density of solid > density of water
- Does not change if density of solid < density of water
- Relative density is measured by Hydrometer.
- The density of sea water is more than that of normal water. This explains why it is easier to swim in sea water.
- When ice floats in water, its 1/10 the part remain outside the water.
- If ice floating in water in a vessel melts, the level of water in the vessel does not change.
- Purity of milk is measured by lactometer.
Surface Tension
- Surface tension is the property of a liquid by virtue of which it has the tendency to have the area of its free surface minimum as if it were under tension like a stretched elastic membrane.
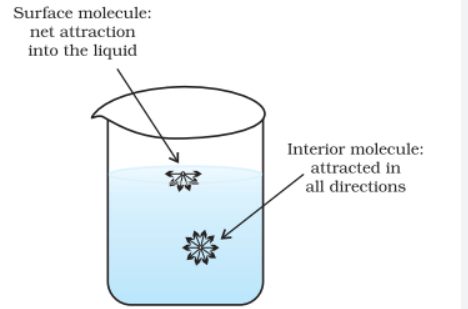
- Surface tension of a liquid decreases with the increase of temperature and becomes zero at critical temperature.
- Surface tension of a liquid is measured by the normal force acting per unit length on either side of an imaginary line drawn on the free surface of liquid and tangential to the surface.
Applications
- A liquid drop attains spherical shape due to surface tension as for given volume, sphere has minimum surface area.
- If a clean and dry needle is kept slowly on the surface of water, it floats due to surface tension.
- The addition of detergent or soap decrease the surface tension of water and thus increases the cleaning ability.
- Bubbles of soap solution are big because addition of soap decreases the surface tension of water.
- When kerosene oil is sprinkled on water, the surface tension of water decreases. As a result the larva of mosquitoes floating on the surface of water die due to sinking.
- Warm soup is tasty because at high temperature its surface tension is low and consequently the soup spreads on all parts of the tongue.
Capillarity
- Capillary Tube – A tube having very narrow (fine) and uniform bore is called a capillary tube.
- Capillarity – If a capillary tube is dipped in a liquid, liquid ascends or descends in the capillary tube. This phenomenon is called capillarity. The height by which liquid ascends or descends in a capillary tube depends upon the radius of the tube.
- The capillarity depends on the nature of liquid and material of the tube.
- The liquid which wets the wall of tube rises in the tube and the liquid which does not wet the wall of tube descends in the tube.
- For example, when a glass capillary tube is dipped in water, water rises in the tube and shape of water meniscus is concave, similarly when a glass capillary tube is dipped in mercury, mercury descends in the tube and shape of mercury meniscus is convex.
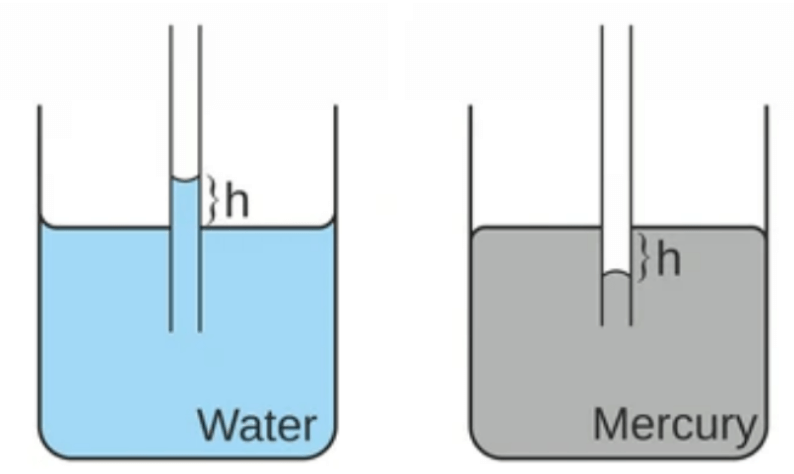
Applications
- A piece of blotting paper soaks ink because the pores of the blotting paper serve as capillary tubes.
- The oil in the wick of a lamp rises due to capillary action of threads in the wick.
- The root hairs of plants draws water from the soil through capillary action.
- To prevent loss of water due to capillary action, the soil is loosened and split into pieces by the farmers.
- If a capillary tube is dipped in water in an artificial satellite, water rises up to other end of tube because of its zero apparent weight, regardless of the length of the tube.
- Action of towel in soaking up water from the body is due to capillary action of cotton in the towel.
- Melted wax, in a candle rises up to wick by capillary action.
Note:-
- Cohesive Force – The force of attraction between the molecules of same substance is called cohesive force. Cohesive force is maximum in solids. This is why solids have a fixed shape. Cohesive force is negligible in case of gases.
- Adhesive Force – Force of attraction between the molecules of different substances is called adhesive force. Due to adhesive force, one body sticks to other.
Viscosity
- Viscous force – The force which opposes the relative motion between different layers of liquid or gases is called viscous force.
- Viscosity is the property of a liquid by virtue of which it opposes the relative motion between its different layers.
- Viscosity is the property of liquids and gases both.
- The viscosity of a liquid is due to cohesive force between its molecules.
- The viscosity of a gas is due to diffusion of its molecules from one layer to other layer.
- Viscosity of gases is much less than that of liquids. Solids do not flow under ordinary conditions so viscosity is not defined for solids.
- Viscosity of an ideal fluid is zero.
- With rise in temperature, viscosity of liquids decreases and that for gases increases.
Bernoulli’s theorem
- According to Bernoulli’s theorem, in case of streamline flow of incompressible and non viscous fluid (ideal fluid) through a tube, total energy (sum of pressure energy, potential energy and kinetic energy) per unit volume of fluid is same at all points. This theorem is simply the principle of conservation of energy.
- Venturimeter, a device used to measure rate of flow of fluid, works on Bernoulli’s theorem.
Elasticity and Wave Motion
Elasticity:
- Elasticity is the property of material of a body by virtue of which the body acquires its original shape and size after the removal of deforming force.
- Elastic Limit: Elastic limit is the maximum value of deforming force upto which a material shows elastic property and above which the material looses its elastic property.
- Stress: The restoring force per unit area set up inside the body subjected to deforming force is called stress.
- Strain: The relative change in dimension or shape of a body which is subjected to stress is called strain. It is measured by ratio of change in length to the original length (longitudinal strain), change in volume to original volume (volume strain).
- Hooke’s law: Under elastic limit, stress is proportional to strain i.e. stress ∝ strain.
Wave
- Wave – A wave is a disturbance that propagates energy from one place to another without the transport of matter.
- Waves are broadly of two types:
- Mechanical Wave: The waves which require material medium (solid, liquid or gas) for their propagation are called mechanical wave or elastic wave. Mechanical waves are of two types:
- Longitudinal Wave: If the particles of the medium vibrate in the direction of propagation of wave, the wave is called longitudinal wave. Waves on springs or sound waves in air are examples of longitudinal waves.
- Transverse Wave: If the particles of the medium vibrate perpendicular to the direction of propagation of wave, the wave is called transverse wave. Waves on strings under tension, waves on the surface of water are examples of transverse waves.
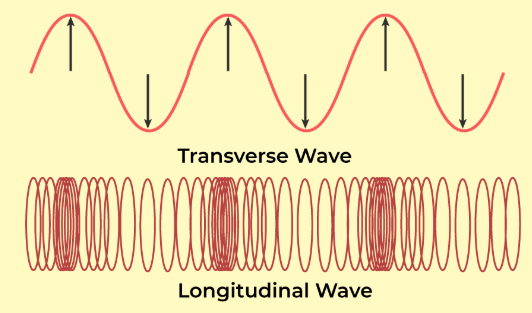
- Waves produced by a moving motor boat on the surface of water is both transverse as well as longitudinal.
- Non-mechanical waves or electromagnetic waves: The waves which do not require medium for their propagation i.e. which can propagate even through the vacuum are called non mechanical waves
- Light, heat are the examples of non-mechanical wave. In fact all the electromagnetic waves are non-mechanical
- All the electromagnetic wave consists of photon.
- In electromagnetic wave, electric field vector and magnetic field vector vibrate perpendicular to each other and also perpendicular to the direction of propagation of wave.
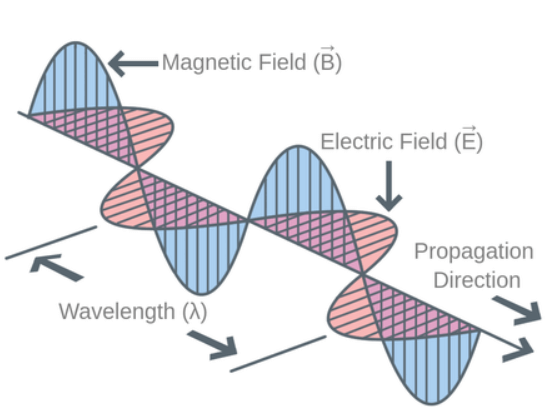
Properties of electromagnetic waves
- They are neutral (uncharged).
- They propagate as transverse wave.
- They propagate with the velocity of light.
- They contains energy and momentum.
| Electro-magnetic Waves | Discoverer | Wavelength range (in meter) | Frequency range |
| γ-Rays | Henry Becquerel | 10⁻¹⁴ to 10⁻¹⁰ | 10²⁰ to 10¹⁸ |
| X-Rays | W. Roentgen | 10⁻¹⁰ to 10⁻⁸ | 10¹⁸ to 10¹⁶ |
| Ultraviolet rays | Ritter | 10⁻⁸ to 10⁻⁷ | 10¹⁶ to 10¹⁴ |
| Visible | Newton | 3.9×10⁻⁷ to 7.8×10⁻⁷ | 10¹⁴ to 10¹² |
| Infrared rays | Hershel | 7.8×10⁻⁷ to 7.8×10⁻³ | 10¹² to 10¹⁰ |
| radio waves | Heinrich Hertz | 10⁻³ to 1 | 10¹⁰ to 10⁸ |
| Long Radio Waves | Marconi | 1 to 10⁴ | 10⁸ to 10⁶ |
- Following waves are not electromagnetic: Cathode rays, Canal rays, α rays, β rays, Sound wave, Ultrasonic wave.
Terms
- Amplitude – Amplitude is defined as the maximum displacement of the vibrating particle on either side from the equilibrium position.
- Wavelength – the distance between any two nearest particle of the medium, vibrating in the same phase. It is denoted by the Greek letter lambda (λ). In transverse wave distance between two consecutive crests or troughs and in longitudinal wave, distance between two consecutive compressions or rarefactions is equal to wavelength.
Sound Wave
- Sound waves are longitudinal mechanical waves.
- According to their frequency range, longitudinal mechanical waves are divided into the following categories:
- Audible or Sound Waves – lie in the frequency range 20 Hz to 20000 Hz are called audible or sound waves. These waves are sensitive to human ear. These are generated by the vibrating bodies such as tuning fork, vocal cords etc.
- Infrasonic Waves – frequencies less than 20 Hz are called infrasonic. These waves are produced by sources of bigger size such as earth quakes, volcanic eruptions, ocean waves and by elephants and whales.
- Ultrasonic Waves – frequencies greater than 20000 Hz are called ultrasonic waves. Human ear can not detect these waves. But certain creatures like dog, cat, bat, mosquito can detect these waves. Bat not only detect but also produce ultrasonic.
- Ultrasonic waves can be produced by Galton’s whistle or Hartmant’s generator or by the high frequency vibrations of a quartz crystal under an alternating electric field (Piezo-electric effect) or by the vibrations of a ferromagnetic rod under an alternating magnetic field (Magnetostriction).
Applications of Ultrasonic Waves
- For sending signals.
- For measuring the depth of sea.
- For cleaning cloths, aeroplanes and machinery parts of clocks.
- For removing lamp-shoot from the chimney of factories.
- In sterilizing of a liquid.
- In Ultrasonography.
Speed of Sound
- Speed of sound is different in different mediums. In a medium, the speed of sound basically depends upon elasticitiy and density of medium.
- Speed of sound is maximum in solids and minimum in gases.
- When sound enters from one medium to another medium, its speed and wavelength changes but frequency remains unchanged.
- In a medium, the speed of sound is independent of frequency.
- Effect of pressure on speed of sound – The speed of sound is independent of pressure i.e. speed remains unchanged by the increase or decrease of pressure.
- Effect of Temperature on speed of sound – The speed of sound increases with the increase of temperature of the medium. The speed of sound in air increases by 0.61 m/s when the temperature is increased by 1°C.
- Effect of humidity on speed of sound – The speed of sound is more in humid air than in dry air because the density of humid air is less than the density of dry air.
Characteristics of Sound waves
- Sound waves have the following three characteristics:
- Intensity
- Intensity of sound at any point in space is defined as amount of energy passing normally through a per unit area around that point per unit time. SI Unit of Intensity is watt/m².
- Intensity of sound at a point is: Inversely proportional to the square of the distance of point from the source and is directly proportional to the square of amplitude of vibration, square of frequency and density of the medium.
- Due to intensity, a sound appears loud or faint to the ear. Actually, the sensation of a sound perceived in ear is measured by another term called loudness which depends on intensity of sound and sensitiveness of the ear. Unit of loudness is bel. A practical unit of loudness is decibel (dB) which is equal to 1/10th of bel. Another unit of loudness is phon.
- Pitch
- Pitch is that characteristic of sound which distinguishes a sharp (or shrill) sound from a grave (dull or flat) sound. Pitch depends upon frequency. Higher the frequency, higher will be the pitch and shriller will be the sound. Lower the frequency, lower will be the pitch and graver will be the sound.
- Quality
- Quality is that characteristic of sound which enables us to distinguish between sounds produced by two sources having the same intensity and pitch. The quality depends upon number, frequency and relative intensities of overtones.
Multiple Reflection of Sound
- When sound waves are reflected again and again (not just once) from surfaces and travel inside a medium, it is called multiple reflection of sound.
- Examples:
- Stethoscope – The sound of heartbeat/lungs reflects many times inside the tube, so the doctor hears it clearly.
- Loudspeaker / Megaphone / Horn / Trumpet – Multiple reflections direct the sound in one direction, making it louder.
- Shehnai / Flute – Sound waves reflect repeatedly inside the instrument to produce music.
- Sound boards in auditoriums – Set up to send sound clearly to the entire hall.
Echo
- The sound waves received after being reflected from a high tower or mountains is called echo.
- To hear echo, the minimum distance between the observer and reflector should be 17 m (16.6 m).
- Persistence of the ear (effect of sound on ear) is 1/10 sec.
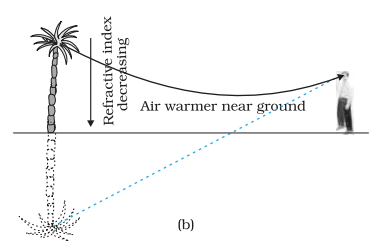
- Due to refraction, sound is heard over longer distances at night than during the day.
Resonance
- If the frequency of imposed periodic force is equal to the natural frequency of a body, the body oscillates with a very large amplitude. This phenomenon is called resonance.
- Examples – Soldiers are asked to break their steps while crossing a bridge to prevent resonance.
Interference of sound
- The modification or redistribution of energy at a point due to superposition of two (or more) sound waves of same frequency is called interference of sound.
- If two waves meet at a point in same phase, intensity of sound is maximum at that point. Such type of interference is called constructive interference.
- Similarly, if the two waves meet at a point in opposite phase, intensity of sound at that point is minimum.
- Such type of interference is called destructive interference.
Diffraction of sound
- Wavelength of sound is of the order of 1 m. If an obstacle of that range appears in the path of sound, sound deviates at the edge of obstacle and propagates forward. This phenomenon is called diffraction of sound.
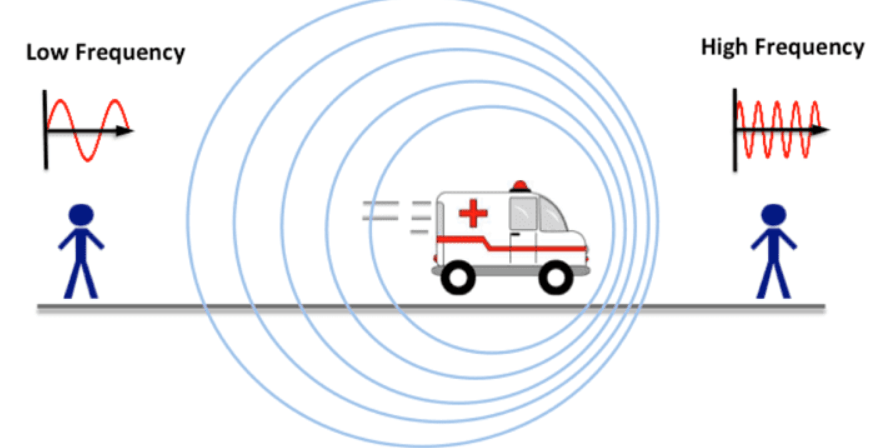
Doppler’s Effect
- If there is a relative motion between source of sound and observer, the apparent frequency of sound heard by the observer is different from the actual frequency of sound emitted by the source. This phenomenon is called Doppler’s effect.
- When the distance between the source and observer decreases, the apparent frequency increases and vice-versa.
Mach Number
- It is defined as the ratio of speed of source of sound to the speed of sound in the same medium under the same condition of temperature and pressure.
- If Mach number > 1, body is called supersonic.
- If mach number > 5, body is called hypersonic.
- If mach number < 1, the body (source) is said to be moving with subsonic speed.
Heat, Temperature and Thermodynamics
Heat
- Heat is that form of energy which flows from one body to other body due to difference in temperature between the bodies. The amount of heat contained in a body depends upon the mass of the body.
- Units of Heat
- C.G.S. unit (calorie) – It is the amount of heat required to raise the temperature of 1 g of pure water by 1°C.
- International calorie – It is the amount of heat required to raise the temperature of 1 g of pure water from 14.5°C to 15.5°C.
- F.P.S. unit : B.Th.U. (British Thermal Unit) – It is the amount of heat required to raise the temp. of 1 pound of pure water by 1°F.
Temperature
- Temperature is that physical cause which decides the direction of flow of heat from one body to other body. Heat energy always flows from body at higher temperature to body at lower temperature.
- Thermometer: The device which measures the temperature of a body is called thermometer.
- Freezing point (F.P.) of mercury is –39°C. Hence to measure temperature below this temperature, alcohol thermometer is used. F.P. of alcohol is –115°C.
Range of different thermometers
- Mercury Thermometer: from –30°C to 350°C
- Constant volume gas thermometer: from –200°C to 500°C (with H₂), below –200°C upto –268°C (with He), above 1000°C upto 1600°C (with N₂ gas and bulb of glazed porcelain)
- Platinum resistance thermometer: from –200°C to 1200°C
- Thermocouple thermometer: from –200°C to 1600°C
Total Radiation (Pyrometer)
- When a body is at high temperature, it glows brightly and the colour of radiation emitted by the body is directly proportional to the fourth power of absolute temperature of the body.
- Its operation relies on the Stefan-Boltzmann Law.
- The Stefan-Boltzmann Law states that the total energy radiated per unit surface area of a black body is directly proportional to the fourth power of its absolute temperature (T^{4})
- This thermometer is not put in contact with the body. But it can not measure temperature below 800°C because at lower temperature emission of radiation is very small and can not be detected.
Specific Heat Capacity
- Specific heat capacity of a material is the amount of heat required to raise the temperature of unit mass of substance through 1°C. Its SI unit is Joule/kilogram kelvin (J/kgK).
- One calorie of heat is required to raise the temperature of 1 gram of water through 1°C. Hence specific heat capacity of water is 1 cal/gram °C.
Thermal Expansion
- When a body is heated its length, surface area and volume increase. The increase in length, area and volume with the increase in temperature is measured in terms of coefficient of linear expansion or linear expansivity (α), coefficient of superficial expansion or superficial expansivity (β) and coefficient of cubical expansion or cubical expansivity (γ).
- Relation between α, β and γ
- α : β : γ = 1 : 2 : 3 or, β = 2α and γ = 3α
Anomalous expansion of water
- Almost every liquid expands with the increase in temperature. But when temperature of water is increased from 0°C to 4°C, its volume decreases.
- If the temperature is increased above 4°C, its volume starts increasing. Clearly, density of water is maximum at 4°C.
Transmission of Heat
- The transfer of heat from one place to other place is called transmission of heat. There are three modes of heat transfer
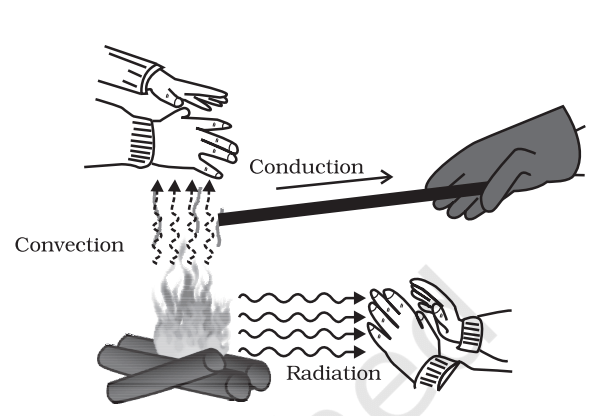
Conduction
- In this process, heat is transferred from one place to other place by the successive vibrations of the particles of the medium without bodily movement of the particles of the medium. In solids, heat transfer takes place by conduction.
- Metals are in general good conductor of heat where as brick, concrete, felt, ice, wood, saw dust, paper, air, glass, cloth are bad conductors of heat.
- This is why ice is packed in saw dust to prevent it from melting.
Convection
- In this process, heat is transferred by the actual movement of particles of the movement from one place to other place. Due to movement of particles, a current of particles is set up which is called convection current.
- In liquids and gases, heat transfer takes place by convection. Earth’s atmosphere is heated by convection.
- Radiation
- In this method transfer of heat takes place with the speed of light without affecting the intervening medium.
Newton’s law of cooling
- The rate of loss of heat by a body is directly proportional to the difference in temperature between the body and its surrounding.
Kirchhoff’s law
- According to Kirchhoff’s law, the ratio of emissive power to absorptive power is same for all surfaces at the same temperature and is equal to emissive power of black body at that temperature.
- Kirchhoff’s law signifies that good absorbers are good emitter.
- If a shining metal ball with some black spot on its surface is heated to a high temperature and seen in dark, the shining metal ball seems dull but the black spot shines brilliantly because the black spot absorbs radiation during heating and emits it in dark.
Stefan’s law
- The radiant energy emitted by a black body per unit area per unit time (i.e. emissive power) is directly proportional to the fourth power of its absolute temperature. E = σT^4 where σ is a constant called Stefan’s constant.
Change of State
- Any material can remain in any of its three states (solid, liquid and gas). To change the substance from one state to other state is called change of state. For this either substance is heated or heat is extracted from the substance. Change of state takes place at a fixed temp.
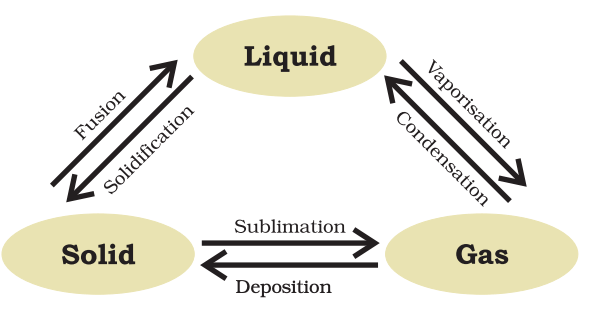
- Fusion – The process by which a substance is changes from solid state to liquid state is called fusion. Fusion takes place at a fixed temperature called melting point (M.P.).
- Freezing(Solidification) – The process by which a substance is changed from liquid state to solid state is called freezing. Freezing takes place at a fixed temperature called freezing point (F.P.). For a substance M.P. = F.P.
- M.P. of a substance changes with the change in pressure.
- Melting point of substances which contracts in the process of fusion (as ice) decreases with the increase in pressure.
- Melting point of substances which expands in the process of fusion (as wax) increases with the increase in pressure.
- With the addition of impurity (as salt in ice), melting point of a substance decreases.
- Vapourisation – The process by which a substance is changed from liquid state to vapour state is called vapourisation. Vapourisation takes place by two methods: Evaporation, Boiling
- Evaporation – The process of vapourisation which takes place only from the exposed surface of liquid and that at all temperatures is called evaporation. Evaporation causes cooling. This is why water in a earthed pot gets cooled in summer.
- Boiling – The process of vapourisation which takes place at a fixed temperature and from whole part of liquid is called boiling. The temperature at which boiling takes place is called boiling point. Boiling point of a liquid increases with the increase in pressure. Boiling point of a liquid increases with the addition of impurity.
- Condensation – The process by which a substance is changed from vapour state to liquid state is called condensation.
Latent heat or heat of transformation
- The amount of heat required to change the state of unit mass of substance at constant temperature is called latent heat.
- If Q heat is required to change the state of a substance of mass m at constant temperature and L is the latent heat, then Q=mL.
- S.I. unit of latent heat is Joule/kilogram.
- Steam produces more severe burn than water at same temperature because internal energy of steam is more than that of water at same temperature (due to latent heat).
- Any material has two types of latent heat.
- Latent heat of fusion
- It is the amount of heat energy required to convert unit mass of a substance from solid state to liquid state at its melting point without any change in temperatures. It is also the amount of heat released by the unit mass of a liquid when it changes into solid state at its freezing point. Latent Heat of fusion – 80 Cal/g
- Latent heat of vapourisation
- It is the amount of heat required to change the unit mass of a substance from liquid state to vapour state at its boiling point. It is also the amount of heat released when the unit mass of a vapour is changed into liquid. Latent Heat of vapourisation – 540 Cal/g
- Latent heat of fusion
Sublimation
- Sublimation is the process of conversion of a solid directly into vapour.
- Sublimation takes place when boiling point is less than the melting point.
- Sublimation is shown by camphor or ice in vacuum.
Hoar Frost
- Hoar frost is just the reverse process of sublimation i.e. it is the process of direct conversion of vapour into solid.
Relative Humidity
- Relative humidity is defined as the ratio of the amount of water vapour present in air to the maximum amount of water vapour that air can hold at the same temperature.
- The ratio is multiplied by 100 to express the relative humidity in percentage.
- Relative humidity is measured by Hygrometer.
- Relative humidity increases with the increase in temperature.
Thermodynamics
First law of thermodynamics
- Heat energy given to a system is used in the following two ways:
- In increasing the temperature and hence internal energy of the system.
- In doing work by the system. If ΔQ = Heat energy given to the system, ΔU = Increase in the internal energy of the system and ΔW = work done by the system. Then ΔQ=ΔU+ΔW.
- Internal energy of a system consist of the potential and kinetic energies of the molecules of the system associated with the mutual interactions and random motion of its molecules.
- First law of thermodynamics is equivalent to principle of conservation of energy.
- Isothermal Process – If the changes are taking place in a system in such a way that temperature of the system remains constant throughout the change, then the process is said to be isothermal.
- Adiabatic Process – If the changes are taking place in a system in such a way that there is no exchange of heat energy between the system and the surrounding, then the process is said to be adiabatic process. If carbon dioxide is suddenly expanded, it is changed into dry ice. This is an example of adiabatic process.
Second law of Thermodynamics
- It explains natural processes’ direction (towards more disorder/entropy), why heat flows hot-to-cold, and sets limits on engine efficiency (100% impossible). Key concepts include Entropy (S) as disorder, irreversibility (e.g., ice melting), energy quality (degrades)
- Kelvin’s statement : Whole of the heat can never be converted into work.
- Clausius statement : Heat by itself can not flow from a body at lower temperature to body at higher temperature.
Light and Optics
Light
- Light is a form of energy which is propagated as electromagnetic waves. In the spectrum of electromagnetic waves it lies between ultra-violet and infra-red region and has wavelength between 3900 Å to 7800 Å.
- Electromagnetic waves are transverse, hence light is transverse wave.
- All the electromagnetic waves including light do not require medium to propagate.
- Wave nature of light explains rectilinear propagation, reflection, refraction, interference, diffraction and polarisation of light.
- The phenomena like photoelectric effect, compton effect are not explained on the basis of wave nature of light. These phenomena are explained on the basis of quantum theory of light (particle nature of light as proposed by Einstein).
- In quantum theory, light is regarded as a packet or bundle of energy called photon. Photon is associated with energy E where E=hν
- Clearly light behaves as wave and particle both. Thus light has dual nature.
- Speed of light is maximum in vacuum and air (3 × 10⁸ m/s). Light takes 8 minute 19 second (499 second) to reach from sun to earth. The light reflected from moon takes 1.28 second to reach earth.
Refractive index
- R.I. of a medium is defined as the ratio of speed of light in vacuum to the speed of light in the medium. Speed of light is different in different media. Velocity of light is large in a medium which has small refractive index.
Reflection of light
- Light moving in one medium when falls at the surface of another medium, part of light returns back to the same medium. This phenomenon of returning back of light in first medium at the interface of two media is known as reflection of light.
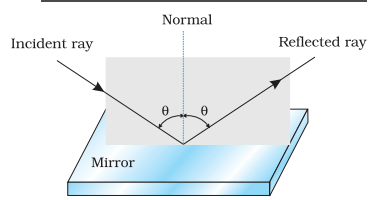
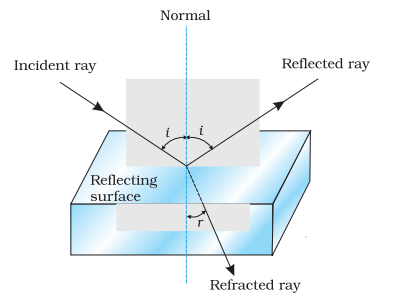
Laws of reflection
- The incident ray, reflected ray and the normal all lie in the same plane.
- The angle of reflection is equal to the angle of incidence.
- Reflection from plane mirror
- The image is virtual, laterally inverted. The size of image is equal to that of object.
- The distance of image from the mirror is equal to distance of object from the mirror. If an object moves towards (or away from) a plane mirror with speed v, the image moves towards (or away) with a speed 2v relative to the object.
- Focal length of a plane mirror is infinite.
- To see his full image in a plane mirror, a person requires a mirror of at least half of his height.
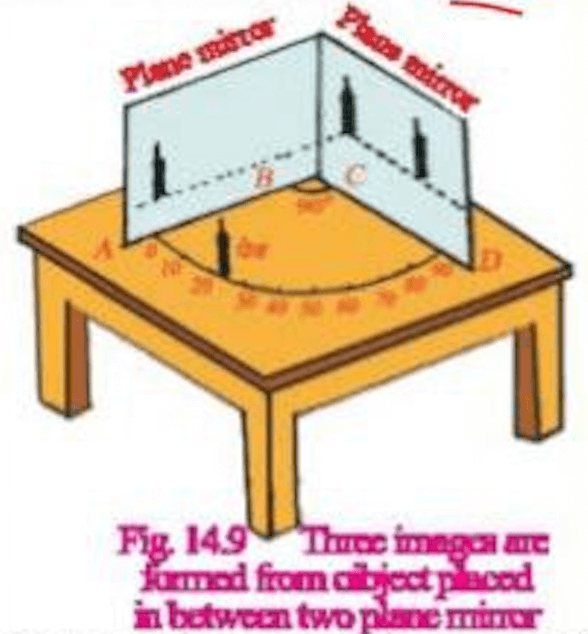
- If two plane mirrors are inclined to each other at an angle 𝜃 then the number of images of a point object formed are determined as follows
- If 360/ θ is even integer = Then number of image formed (360/θ – 1)
- If 360/ θ is odd integer than number of images will be 360/θ – 1
- If the object is metrically placed and number of images is 360/θ if the object is not symmetrically placed.
- Object is placed between two parallel plane mirrors

Object is placed between two perpendicular plane mirrors
- Reflection from spherical mirror
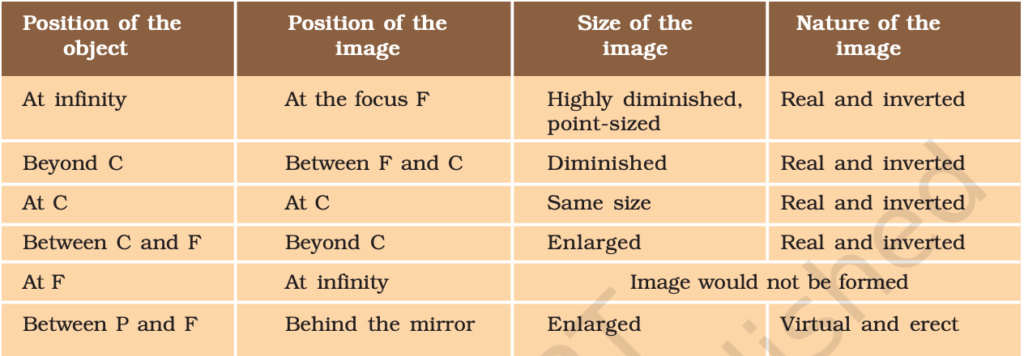
- Position & nature of image formed by a spherical mirror
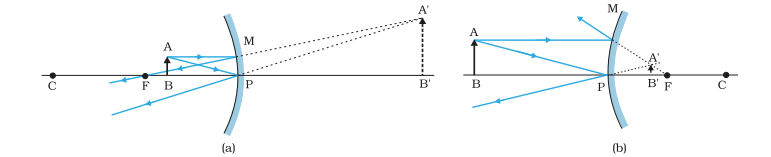
Concave mirror
Uses of Concave mirror
- As a shaving glass.
- As a reflector for the headlights of a vehicle, search light.
- In ophthalmoscope to examine eye, ear, nose by doctors.
- In solar cookers.
- In concentrating light at a given spot.
Convex mirror
| Position of object | Position of image | Size of image | Nature of image |
| At infinity | At Focus | Highly diminished | Virtual, erect |
| In front of mirror | Between pole and focus | Diminished | Virtual, erect |
Note: Image formed by a convex mirror is always virtual, erect and diminished.
Uses of Convex mirror
- As a rear view mirror in vehicle because it provides the maximum rear field of view and image formed is always erect.
- In sodium reflector lamp.
Refraction of light.
- When a ray of light propagating in a medium enters the other medium, it deviates from its path. This phenomenon of change in the direction of propagation of light at the boundary when it passes from one medium to other medium is called refraction of light.
- When a ray of light enters from rarer medium to denser medium (as from water to glass), it deviates towards the normal drawn on the boundary of two media at the incident point. Similarly in passing from denser to rarer medium, a ray deviates away from the normal. If light is incident normally on the boundary i.e. parallel to normal, it enters the second medium undeviated.
Laws of refraction
- Incident ray, refracted ray and the normal lie in the same plane.
- Snell’s law: For a given colour of light, the ratio of sine of angle of incidence to the sine of angle of refraction is a constant, is called refractive index of second medium with respect to the first medium.
- Absolute refractive index of a medium is defined as the ratio of speed of light in free space (vacuum) to that in the given medium. i.e. absolute refractive index (μ)
- The refractive index of a medium is different for different colours. The refractive index of a medium decreases with the increase in wavelength of light. Hence refractive index of a medium is maximum for violet colour of light and minimum for red colour of light.
- The refractive index of a medium decreases with increase in temperature. But this variation is very small.
- When a ray of light enters from one medium to other medium, its frequency and phase do not change but wavelength and velocity change.
Some illustrations of Refraction
- Bending of a linear object when it is partially dipped in a liquid inclined to the surface of the liquid.
- Twinkling of stars.
- Oval shape of sun in the morning and evening.
- An object in a denser medium when seen from a rarer medium appears to be at a smaller distance. This is why
- (a) A fish in a pond when viewed from air appears to be at a smaller depth than actual depth
- (b) A coin at the base of a vessel filled with water appears raised
- (c) Water tank appears shallower when viewed from the top.
The refractive index is proportional to wavelength. So shorter the wavelength, the less it refracts (deviates). Hence if a glass slab is place over coloured letters, violet letter having shortest wavelength will appear less raised than any other colour. Red colour being of longest wavelength will appear most raised.
Critical angle
- In case of propagation of light from denser to rarer medium through a plane boundary, critical angle is the angle of incidence for which angle of refraction is 90°.
Total Internal Reflection
- If light is propagating from denser medium towards the rarer medium and angle of incidence is more than critical angle, then the light incident on the boundary is reflected back in the denser medium, obeying the laws of reflection. This phenomenon is called total internal reflection as total light energy is reflected, no part is absorbed or transmitted.
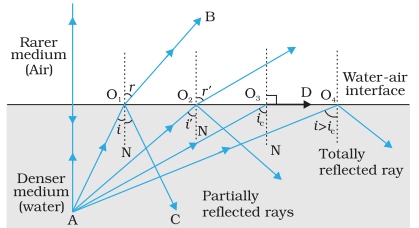
For total internal reflection:
- Light must be propagating from denser to rarer medium. This is why if light goes from water to glass, total internal reflection will not take place.
- Angle of incidence must exceeds the critical angle.
Illustrations of total internal reflection
- Sparkling of diamond
- Mirage and looming
- Shining of air bubble in water
- Increase in duration of sun’s visibility – The sun becomes visible even before sunrise and remains visible even after sunset due to total internal reflection of light
- Shining of a smoked ball or a metal ball on which lamp soot is deposited when dipped in water.
- Optical Fibre: Optical fibre consists of thousands of strands of a very fine quality glass or quartz (of refractive index 1.7), each strand coated with a layer of material of lower refractive index (1.5). In it light is propagated along the axis of fibre through multiple total internal reflection, even though the fibre is curved, without loss of energy.
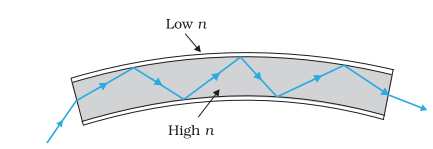
- Applications :
- For transmitting optical signals and the two dimensional pictures.
- For transmitting electrical signals by first converting them to light.
- For visualising the internal sites of the body by doctors in endoscopy.
Refraction of Light Through Lens
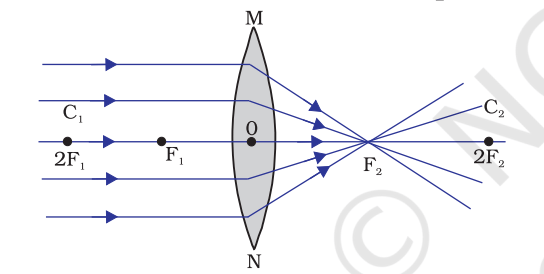
- Convex lens – When a lens is thicker at the middle than at the edges, it is called a convex lens or a converging lens.
- Concave lens – When the lens is thicker at the edges than in the middle, it is called as concave lens or diverging lens.
- Power of a lens – Power of a lens is its capacity to deviate a ray. It is measured as the reciprocal of the focal length in meters. SI Unit of power is dioptre (D). Power of a convex lens is positive and that of a concave lens is negative. If two lenses are placed in contact, then the power of combination is equal to the sum of powers of individual lenses.
Formation of images by lenses
Convex Lens
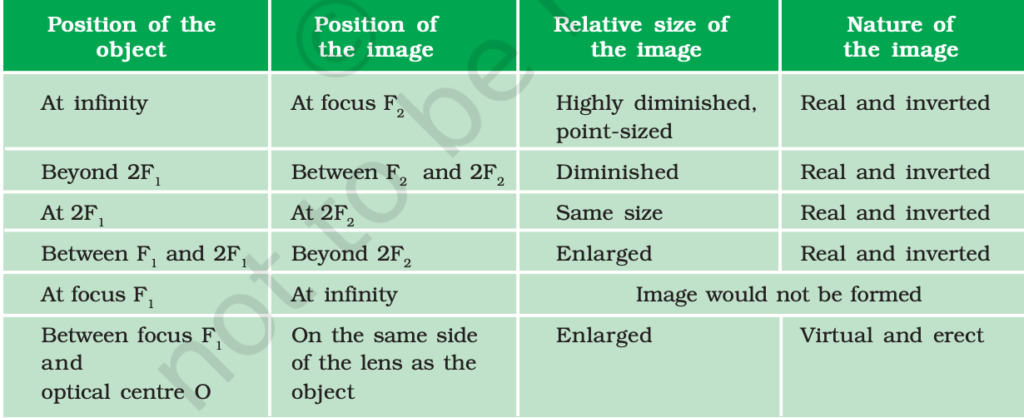
Concave lenses
| Position of object | Position of image | Size of image | Nature of image |
| At infinity | At focus | Highly diminished | Virtual and erect |
| Between lens and infinity | Between lens and F on the same side | Diminished | Virtual and erect |
Dispersion of Light
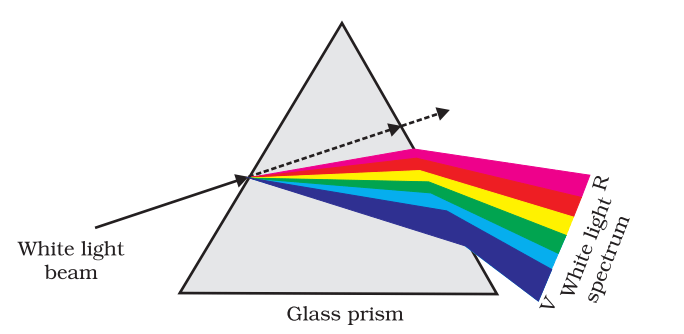
- When a ray of white light (or a composite light) is passed through a prism, it gets splitted into its constituent colours. This phenomenon is called dispersion of light. The coloured pattern obtained on a screen after dispersion of light is called spectrum.
- The dispersion of light is due to different deviation suffered by different colours of light. The deviation is maximum for violet colour and minimum for red colour of light. The different colours appeared in the spectrum are in the following order, violet, indigo, blue, green, yellow, orange and red. (VIBGYOR)
- The dispersion of light is due to different velocities of light of different colours in a medium. As a result, the refractive index of a medium is different for different colours of light.
- The velocity of light in a medium is maximum for that colour for which refractive index is minimum. Clearly, the velocity of violet colour of light is minimum in a medium and refractive index of that medium is maximum for violet colour. Similarly, the velocity of light in a medium is maximum for red colour and refractive index of that medium is minimum for red colour.
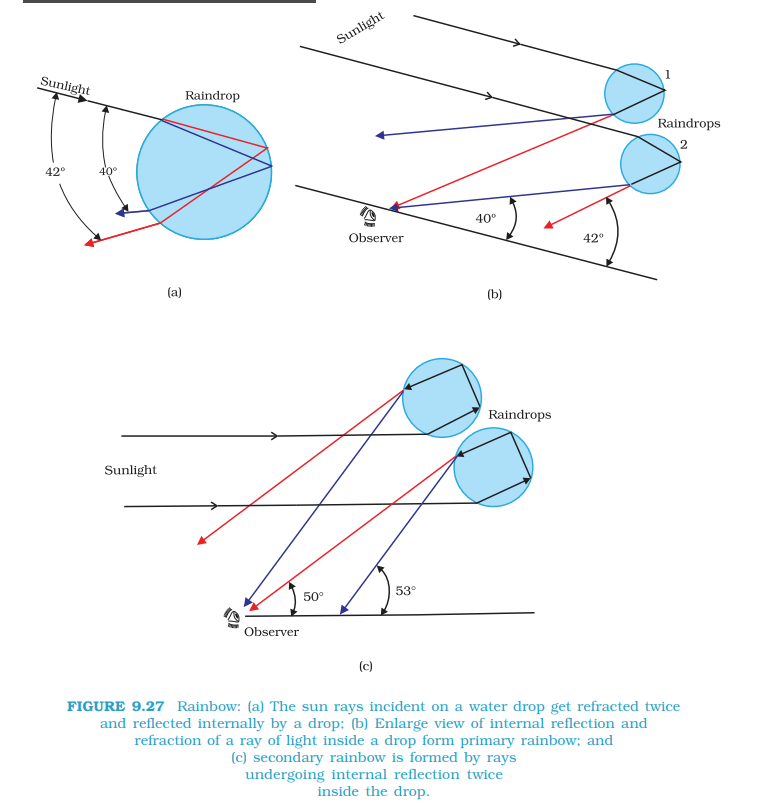
Rainbow
- Rainbow is the coloured display in the form of an arc of a circle hanging in the sky observed during or after a little drizzle appearing on the opposite side of sun. Rainbow is formed due to dispersion and refraction of sun light by the suspended water droplets.
- Rainbow is of two types:
- Primary rainbow is formed due to two refractions and one total internal reflection of light falling on the raindrops. In the primary rainbow, the red colour is on the convex side and violet on the concave side. Primary rainbow has an angular width of 2° at an average angle of elevation of 41°.
- Secondary rainbow is formed due to two refractions and two internal reflections of light falling on rain drops. The order of colour on the secondary rainbow is in the reverse order and has an angular width of 3.5° at an average elevation of 52.75°. Secondary rainbow is less intense than primary rainbow.
Theory of Colours
- Colour is the sensation perceived by the cones in the eye due to light.
- Primary Colours – The spectral colours blue, green and red are called primary colours because all the colours can be produced by mixing these in proper proportion. Blue + Red + Green = White
- Secondary Colours
- The colour produced by mixing any two primary colours is called a secondary colour. There are three secondary colours yellow, magenta and cyan(Blue + Green)
Scattering of light
- When light waves fall on small particles such as dust, water droplets, suspended particles in colloidal solution, they are scattered in all directions.
- Scattering of light is maximum in case of violet colour and minimum in case of red colour of light.
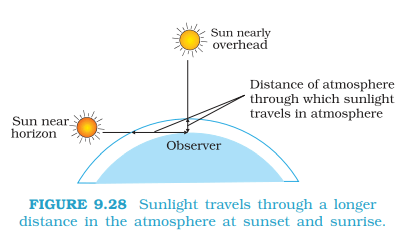
- The blue colour of the sky is due to the scattering of light.
- The brilliant red colour of rising and setting sun is due to scattering of light.
- It is the scattering of light by particles much smaller than the wavelength of light.
- Works mainly for molecules and very small particles (like O₂, N₂ in the atmosphere).
- Intensity of scattering depends strongly on wavelength. Mathematical Relation = Intensity (I) ∝ 1/λ⁴
- Scattering of light is inversely proportional to the fourth power of the wavelength.
- Shorter wavelength → more scattering.
- Longer wavelength → less scattering.
- That’s why violet & blue light scatter more than red light.
- Applications / Natural Phenomena Explained
- Blue colour of the sky – because blue light scatters more than red.
- Reddish sun at sunrise & sunset – at low sun angle, blue is scattered away, only red/orange reach our eyes.
- Why sky is white in polluted areas – bigger dust particles scatter all colours → whitish appearance (Mie scattering)
- Visibility of distant mountains (bluish tint) – blue light scattered from particles in the air.
Interference of light
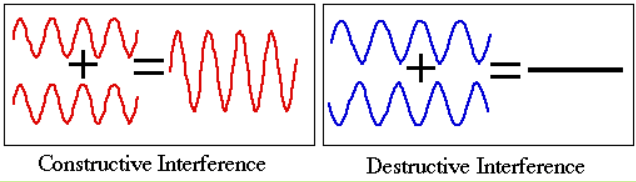
- When two light waves of exactly the same frequency and a constant phase difference travel in same direction and superimpose then the resultant intensity in the region of superposition is different from the sum of intensity of individual waves. This modification in the intensity of light in the region of superposition is called interference of light. Interference is of two types:
- Constructive interference – At some points, where the two waves meet in same phase, resultant intensity is maximum. Such interference is called constructive interference.
- Destructive interference – At some points, where the two waves meet in opposite phase, resultant intensity is minimum. Such interference is called destructive interference.
Diffraction of light
- Diffraction of light is the phenomenon where light waves bend and spread out as they pass around the edges of obstacles or through narrow openings, moving into the geometrical shadow area, which happens because light behaves as a wave.
- The spreading of light energy beyond the limit prescribed by rectilinear propagation of light is called diffraction of light. In other words, diffraction is the process by which a beam of light or other systems of waves is spread out as a result of passing through a narrow opening or across an edge.
Polarisation of light
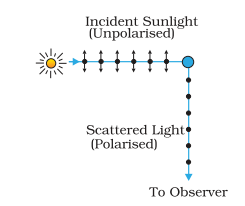
- Polarisation is the only phenomenon which proves that light is a transverse wave.
- Light is an electromagnetic wave in which electric and magnetic field vectors vibrate perpendicular to each other and also perpendicular to the direction of propagation.
- Polarisation is the phenomenon of restricting the vibrations of a light in a particular direction in a plane perpendicular to the direction of propagation of wave.
Electricity and Magnetism
Static Electricity
- When two bodies are rubbed together, they acquire the property of attracting light objects like small bits of paper, dust particles etc. The bodies which acquire this property are said to be electrified or charged with electricity.
- Charge: Charge is the basic property associated with matter due to which it produces and experiences electrical and magnetic effects.
- Benjamin Franklin named the two types of charges as positive and negative.
- Similar charges repel each other and opposite charges attract each other.
- Charging of bodies takes place due to transfer of electrons from one body to another body.
- Surface density of charge – defined as the amount of charge per unit area on the surface of conductor.
- The surface density of charge at a point on the surface of conductor depends upon the shape of conductor and presence of other conductors or insulators near the given conductor.
- The surface density of charge at any part of the conductor is inversely proportional to the radius of curvature at that point.
- This is why surface density of charge is maximum at the pointed parts of the conductor.
Coulomb’s law
- According to Coulomb’s law, the force of attraction or repulsion between two point charges at rest is directly proportional to the product of the magnitudes of the charges and inversely proportional to the square of the distance between them. This force acts on the line joining the two charges.
- Terms
- Electric Field – Region in space around a charge or charged body in which its electrical effect can be experienced is called the electric field of the charge.
- Electric Field Intensity – in an electric field, force experienced by a unit positive charge placed at that point.
- Electric Field of hollow conductor – inside a charged hollow conductor is zero. Charge given to such a conductor (or conductor of any shape) remains on its surface only.
- This explains why a hollow conductor acts as an electrostatic shield. It is for this reason that it is safer to sit in a car or bus during lightning.
- Electric Potential – Electric potential at a point in an electric field is the work done in bringing a unit positive charge from infinity to that point. The SI unit of electric potential is volt. It is a scalar quantity.
- Potential Difference – Work done in bringing a unit positive charge from one point to other point is the potential difference between the two points. Its SI unit is volt and is a scalar quantity.
Current Electricity
- Electric current is defined as the rate of flow of charge or charge flowing per unit time interval. Its direction is the direction of flow of positive charge. Its SI unit is ampere (A). It is a scalar quantity.
- A current of one ampere flowing through a conductor means 6.25 × 10¹⁸ electrons are entering at one end or leaving the other end of the conductor in one second.
Resistance
- The opposition offered by a conductor to the flow of current through it is called resistance. It arises due to collisions of drifting electrons with the core ions. Its SI unit is ohm.
Ohm’s law
- If physical conditions like temperature, intensity of light etc. remains unchanged then electric current flowing through a conductor is directly proportional to the potential difference across its ends. If V is the potential difference across the ends of a conductor and I is the current through it, then according to Ohm’s law , V ∝ I or, V = RI.
- The resistance of a conductor is directly proportional to its length and inversely proportional to its cross sectional area i.e. if l and A are respectively length and cross sectional area of a conductor and R is its resistance then
- R ∝ l / A or, R = ρ l / A
Combination of Resistance
- Series combination – In series combination, the equivalent resistance is equal to the sum of the resistances of individual conductors. (R = R₁ + R₂ + ….. Rₙ)
- Parallel combination – In parallel combination, the reciprocal of equivalent resistance is equal to the sum of the reciprocal of individual resistances. (1 / R = 1 / R₁ + 1 / R₂ + …. + 1 / Rₙ)
- Electric Power – The rate at which electrical energy is consumed in a circuit is called electric power. Electric power = I²R = V² / R = VI Its SI unit is watt. 1 kilowatt hour = 3.6 × 10⁶ joule. 1 kWh is also called board of trade unit.
- Ammeter – a device used to measure electric current in a circuit. It is connected in series in the circuit. The resistance of an ideal ammeter is zero.
- Voltmeter – a device used to measure the potential difference between two points in a circuit. It is connected in parallel to the circuit. The resistance of an ideal voltmeter is infinite.
- Electric fuse – a protective device used in series with an electric appliance to save it from being damaged due to high current. In general, it is a small conducting wire of alloy of copper, tin and lead. Pure fuse is made up of tin. The fuse material should be of low melting point and high resistance.
- Galvanometer – a device used to detect and measure small electric current in a circuit. It can measure current up to 10⁻⁶ A.
- Electromagnetic Induction – Whenever there is change of magnetic flux linked with a circuit, an emf is induced in the circuit. This phenomenon is called electromagnetic induction. The emf so developed is called induced emf and the current so generated (if circuit is closed) is called induced current. Direction of induced current is determined with the help of Fleming’s right hand rule or Lenz law.
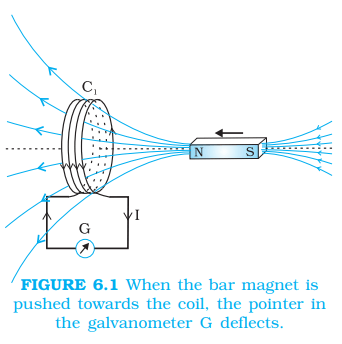
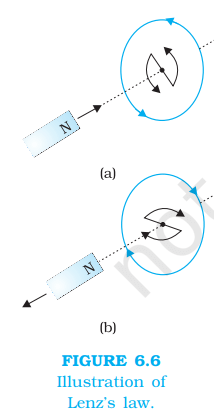
- Lenz law – the direction of an induced electric current (or electromotive force, EMF) in a conductor, caused by a changing magnetic field, always creates its own magnetic field that opposes the original change in magnetic flux that produced it, ensuring energy conservation.
- Transformer– Transformer is a device which converts low voltage A.C. into high voltage A.C. and high voltage A.C. into low voltage A.C. It is based on electromagnetic induction and can be used only in case of alternating current.
Magnetism
- Magnetism is the property displayed by magnets and produced by the movement of electric charges, which results in objects being attracted or pushed away.
- A magnet is characterised
- Attractive property : A magnet attracts magnetic substances like iron, cobalt, nickel and some of their alloys like magnetite (Fe₃O₄).
- Directive property : When a magnet is freely suspended, it aligns itself in the geographical north south direction.
- A magnet may be 1. Natural 2. Artificial
- Natural magnet is oxide of iron. But due to irregular shape, weak magnetism and high brittleness, natural magnets find no use in the laboratory.
- The magnets made by artificial methods are called artificial magnets or man made magnets. They may be of different types like bar magnet, horse shoe magnet, Robinson’s ball ended magnet, magnetic needle, electromagnet etc.
- The middle part of a magnet has minimum magnetic (attracting) power.
- The imaginary line joining the two poles of a magnet is called magnetic axis of the magnet.
- Similar poles repel each other and dissimilar poles attract each other.
- When a magnetic substance is placed near a magnet, it gets magnetised due to induction.
- Magnetic Field – Region in space around a magnet where the magnet exerts its magnetic effect is called magnetic field of the magnet.
- Intensity of magnetic field or magnetic flux density
- Magnetic flux density of a point in a magnetic field is the force experienced by a north pole of unit strength placed at that point. Its SI unit is newton/ampere-meter or weber/meter² or tesla (T).
Magnetic lines of force
- The magnetic lines of force are imaginary curves which represent a magnetic field graphically. The tangent drawn at any point on the magnetic lines of force gives the direction of magnetic field at that point.
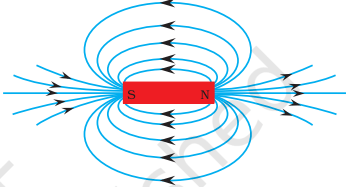
- Properties of magnetic lines of force
- Magnetic lines of force are closed curves. Outside the magnet they are from north to south pole and inside the magnet they are from south to north pole.
- Two lines of force never intersect each other.
- If the lines of force are crowded, the field is strong.
- If the lines of force are parallel and equidistant, the field is uniform.
- Magnetic Substance – divided into three categories.
- Diamagnetic Substance – Substances which when placed in a magnetic field acquire feeble magnetism opposite to the direction of magnetic field. Examples : Bismuth, Zinc, Copper, Silver, Gold, Diamond, Mercury, Water etc.
- Paramagnetic Substance – Substances which when placed in a magnetic field acquire a feeble magnetism in the direction of the field. Examples : Aluminium, Platinum, Manganese, Sodium, Oxygen etc.
- Ferromagnetic Substance – Substances, which when placed in a magnetic field, are strongly magnetised in the direction of the field. Examples : Iron, Cobalt, Nickel etc.
Curie Temperature
- As temperature increases, the magnetic property of ferromagnetic substance decreases and above a certain temperature the substance changes into paramagnetic substance. This temperature is called Curie temperature.
Terrestrial Magnetism
- Our earth behaves as a powerful magnet whose south pole is near the geographical north pole and whose north pole is near the geographical south pole. The magnetic field of earth at a place is described in terms of following three elements.
- Declination : The acute angle between the magnetic meridian and geographical meridian at a place is called the angle of declination at that place.
- Dip or Inclination : Dip is the angle which the resultant earth’s magnetic field at a place makes with the horizontal. At poles and equator, dip is 90° and 0° respectively.
- Horizontal component of earth’s magnetic field : At a place it is defined as the component of earth’s magnetic field along the horizontal in the magnetic meridian. Its value is different at different places. (approximately 0.4 gauss or 0.4 × 10⁻⁴ tesla)
Eddy Currents
- Eddy currents are loops of electric current within conductors that are placed in a changing magnetic flux. They are also named “ Foucault currents”.
- Eddy currents oppose the cause which produces them. When a magnet is brought near a copper plate, the eddy currents produced in the plate will generate their own magnetic field which will in turn push the magnet. This is in accordance with Lenz’s law.
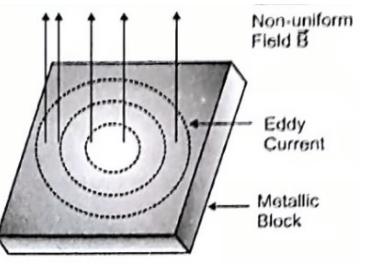
- Applications of eddy currents
- Electromagnetic brakes – They are used in electric trains. Magnetic flux is passed perpendicular to the direction of rotation of the wheels. This produces eddy currents to flow in a direction opposite to that of the rotation of the wheel, thereby retarding their motion and stopping the train.
- Induction motors
Atomic, Nuclear and Modern Physics
Atomic & Nuclear Physics
- Atom is the smallest part of matter which takes part in chemical reactions. Atoms of the same element are similar in mass, size and characteristics. Atom consists of three fundamental particles electron, proton and neutron. All the protons and neutrons are present in the central core of atom called nucleus. Electrons revolve around the nucleus.
- In an atom, electrons and protons are equal in number and have equal and opposite charge. Hence atom is neutral.
Radioactivity
- Radioactivity is the sending out of harmful radiation or particles, caused when atomic nuclei breakup spontaneously.
- Radioactivity was discovered by Henry Becquerel for which he received the Nobel Prize of Physics jointly with Madame Curie and Pierre Curie
- The nuclei having protons 83 or more are unstable. They emit α, β and γ particles and become stable. The elements of such nucleus are called radioactive elements and the phenomenon of emission of α, β and γ particles is called radioactivity. β rays are fast moving electrons. In the nucleus an electron is created due to conversion of neutron into proton. γ rays are electromagnetic waves. γ rays are emitted after the emission of α and β rays.
Properties of α, β and γ particles
| Property | α | β | γ |
| Origin | Nucleus | Nucleus | Nucleus |
| Nature | Positively charged | Negatively charged | Neutral |
| Composition | He⁴ | e⁰ | Photon |
| Mass | 6.4 × 10⁻²⁷ kg | 9.1 × 10⁻³¹ kg | zero |
| Charge | +2e | −e | zero |
| Chemical effect | Affects photographic plate | Affects photographic plate | Affects photographic plate |
| Effect of electric and magnetic field | Deflected | Deflected | No effect |
| Penetrating power | Minimum | In between the other two | Maximum |
| Ionising power | Maximum | In between the other two | Minimum |
| Velocity | Between 1.4 × 10⁷ m/s to 2.2 × 10⁷ m/s | 1% to 99% velocity of light | 3 × 10⁸ m/s |
- With the emission of an α-particle, atomic number is decreased by 2 and mass number is decreased by 4.
- With the emission of a β-particle, atomic number is increased by one and mass number does not change.
- The effect on the mass number and atomic number with the emission of α, β and γ rays is decided by Group-displacement law or Soddy-Fajans Law.
- Radioactivity is detected by G.M. Counter.
- The time in which half nuclei of the element is decayed is called half life of the radioactive substance.
- Radioactive carbon-14 is used to measure the age of fossils and plants. (Carbon dating) In this method age is decided by measuring the ratio of C¹² and C¹⁴.
Nuclear Fission and Fusion
- Nuclear Fission – The nuclear reaction in which a heavy nucleus splits into two nuclei of nearly equal mass is nuclear fission. The energy released in the nuclear fission is called nuclear energy.
- Chain Reaction – When uranium atom is bombarded with slow neutrons, fission takes place. With the fission

- Chain Reaction – When uranium atom is bombarded with slow neutrons, fission takes place. With the fission of each uranium nucleus, on an average 3 neutrons and large amount of energy is released. These neutrons cause further fission. Clearly a chain of fission of uranium nucleus starts which continues till whole of uranium is exhausted. This is called chain reaction.
- Little Boy was a uranium-based atomic bomb, utilizing the nuclear fission of uranium-235 dropped on Hiroshima on 6 august .
- The “Fat Man” atom bomb was the second atomic bomb (plutonium-based) used in World War II, dropped by the United States on Nagasaki, Japan, on August 9, 1945.
- Chain reaction is of the following two types :
- Uncontrolled Chain Reaction – In each fission reaction, three more neutrons are produced. These three neutrons may cause the fission of three other U²³⁵ nuclei producing 9 neutrons and so on. As a result the number of neutron goes on increasing till the whole of fissionable material is consumed. This chain reaction is called uncontrolled or explosive chain reaction. This reaction proceeds very quickly and a huge amount of energy is liberated in a short time.
- Atom bomb – based on nuclear fission. U²³⁵ and Pu²³⁹ are used as fissionable material. This bomb was first used by USA against Japan in second world war (6th August, 1945 at Hiroshima & 9th August, 1945 at Nagasaki).
- Controlled Chain Reaction – A fission chain reaction which proceeds slowly without any explosion and in which the energy released can be controlled is known as controlled reaction. Actually in this situation only one of the neutrons produced in each fission is able to cause further fission.
- Nuclear Reactor or Atomic Pile – Nuclear reactor is an arrangement in which a controlled nuclear fission reaction takes place.
- Components of nuclear reactor :
- Fissionable Fuel : U²³⁵ or U²³⁹ is used.
- Moderator : Moderator decreases the energy of neutrons so that they can be further used for fission reaction. Heavy water and graphite are used as moderator.
- Control rod : Rods of cadmium or boron are used to absorb the excess neutrons produced in fission of uranium nucleus so that the chain reaction continues to be controlled.
- Coolant : A large amount of heat is produced during fission. Coolant absorbs that heat and prevents excessive rise in the temperature. The coolant may be water, heavy water (D₂O—deuterium ( H² ) is an isotope of hydrogen), or a gas like He or CO₂.
Nuclear Fusion
- When two or more light nuclei combine together to form a heavier nucleus, tremendous energy is released. This phenomenon is called nuclear fusion.
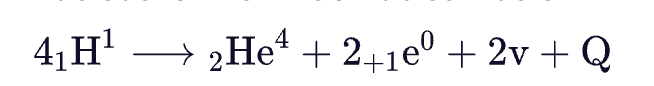
- A typical example of nuclear fusion is : The energy released by sun and other stars is by nuclear fusion. For the nuclear fusion, a temperature of the order of 10⁸ K is required.
- Hydrogen bomb – made by American scientists in 1952. This is based on nuclear fusion. It is 1000 times more powerful than atom bomb.
- Note:-
- Isotopes: These are atoms of the elements having the same atomic number but different mass number. Isotopes of Carbon-¹²C, ¹³C, ¹⁴C (Z = 6, 6, 6)
- In isotopes number of protons and electrons are the same but the number of neutrons are different.
- Some Important Radioactive Isotopes and Their Uses
- Uranium-235 (U²³⁵): Used in the production of nuclear energy
- Sodium-24 (Na²⁴): Used in detecting blood circulation
- Cobalt-60 (Co⁶⁰): Used in the treatment of blood cancer
- Arsenic-74 (As⁷⁴): Used in the diagnosis of tumors
- Iodine-131 (I¹³¹): Used in the treatment of thyroid gland
- Phosphorus-32 (P³²): Used in the treatment of leukemia
- Iron-59 (Fe⁵⁹): Used in diagnosing anemia
- Isobars: These are atoms of the elements having the same mass number but different atomic numbers. E.g. ⁴⁰K (Z=19), ⁴⁰Ca (Z=20), ⁴⁰Ar (Z=18)
- Isotones: These are atoms of different elements having the same number of neutrons. ¹⁴C, ¹⁵N, ¹⁶O ( Z = 6, 7, 8; 8 neutrons in all)
- Isoelectronic : These are atoms/molecules/ions containing the same number of electrons.
- Isomers – chemical compounds that share the same molecular formula but have different arrangements of atoms in space, leading to distinct physical and chemical properties.
- Mass Energy Relation – In 1905 Einstein established the relation between mass and energy on the basis of special theory of relativity. According to this relation, mass can be converted into energy and vice versa.
- 𝐸 = 𝑚𝑐^ 2, where c is the velocity of light and E is the energy equivalent of mass m.
Semiconductor
- Semiconductors are those materials whose electrical conductivity, at room temperature, lies in between that of insulator and conductor. Germanium and silicon are two important semiconductors.
- In a crystal lattice of a semi-conductor, some of the electrons become free from bond formation.
- At the sites of these electrons a deficiency of electrons exists which acts as a virtual positive charge.
- These virtual positive charges are called holes. Semi-conductors are used in the electronics industry.
- Semi-conductors are of two types
- Intrinsic semiconductor – extremely pure form of semiconductor with no doping atoms added.
- Extrinsic semiconductor – chemical impurity is added to increase its conductivity.
| Semiconductor | N type | P type |
| Impurity added | 15th group elements | 13th group elements |
| Charge carriers | majority – electron, minority – hole | Majority hole Minority electron |
- Doping : Adding of chemical impurity to a pure semi conductor is called doping. The amount and type of impurity is closely controlled.
- The electrical conductivity of a semi conductor increases with the increase in temperature.
Science in Daily Life with General Science Facts
Measurement devices
| Devices | Used to measure |
| Aerometer | Air/gas density/weight Measurement |
| Fathometer | Depth of ocean |
| SONAR | Depth of sea bed |
| Pyrheliometer | Solar radiation |
| Anemometer | Wind speed |
| Seismograph | Intensity of earthquake |
| Theodolite | Measuring angular distance in vertical plane and horizontal plane |
| Ammeter | To measure electric current in a circuit |
| Barometer | Atmospheric pressure |
| Pyrometer | Intense temperature |
| Hygrometer | Humidity of atmosphere |
| Sphygmomanometer | To measure blood Pressure |
| Cartometer | To check purity of gold |
| Luxmeter | Intensity of light |
| Odometer | Distance covered by motor vehicles |
| Audiometer | Intensity of sound |
| Lactometer | Purity and density of milk |
| Voltmeter | Electric potential |
| Galvanometer | Presence of electric current in a circuit |
| Geiger muller counter | Radioactivity |
Metals and their ore
| Metal | Ores |
| Silver (Ag) | Ruby Silver, Horn Silver, Galena, Sylvanite, Argentite, Chlorargyrite |
| Manganese (Mn) | Pyrolusite, Magnite, Cryptomallin, Cylomallin |
| Gold (Au) | Calaverite, Silvenites |
| Iron (Fe) | Hematite, Limonite, Magnetite, Siderite, Goethite, Iron Pyrite |
| Zinc (Zn) | Zinc blende, Zincite, Calamine, |
| Mercury (Hg) | Cinnabar |
| Uranium (U) | Carnetite, Pitchblende, Uraninite |
| Lead (Pb) | Galena |
| Tin (Sn) | Cassiterite |
| Copper (Cu) | Cuprite, Copper glance, Copper pyrites, Malachite |
| Magnesium (Mg) | Magnesite, Dolomite, Epsom salt, Kieserite, Carnallite |
| Potassium (K) | Nitre (saltpeter) , Carnallite |
| Aluminium (Al) | Bauxite, Corundum, Felspar, Cryolite, Bauxite, Corundum, Alunite, Kaolin, Kaolinite |
Alloy
| Alloy | Composition |
| Brass | Cu (70%) + Zn (30%) |
| Bronze | Cu (90%) + Sn (10%) |
| German Silver | Cu (60%) + Zn (20%) + Ni (20%) |
| Rolled Gold | Cu (90%) + Al (10%) |
| Gun Metal | Cu (88%) + Sn (10%) + Zn (1%) + Pb (1%) |
| Delta Metal | Cu (60%) + Zn (38%) + Fe (2%) |
| Munz Metal | Cu (60%) + Zn (40%) |
| Dutch Metal | Cu (80%) + Zn (20%) |
| Monel Metal | Cu (70%) + Ni (30%) |
| Rose Metal | Bi (50%) + Pb (28%) + Sn (22%) |
| Solder | Pb (50%) + Sn (50%) |
| Magnallium | Al (95%) + Mg (5%) |
| Duralumin | Al (94%) + Cu (3%) + Mg (2%) + Mn (1%) |
| Type Metal | Sn (5%) + Pb (80%) + Sb (15%) |
| Bell Metal | Cu (80%) + Sn (20%) |
| Stainless Steel | Fe (75%) + Cr (15%) + Ni (10%) + C (0.5%) |
| Nickel Steel | Fe (95%) + Ni (5%) |
Commonly used chemicals in daily life
| Common Name | Chemical Formula |
| Baking soda | NaHCO3 |
| Plaster of paris | CaSO4.1/2 H2O |
| Washing soda | Na2CO3.10 H2O |
| Quick lime | CaO |
| Iron rust | Fe2O3 |
| Bleaching powder | CaOCl2 |
| Milk of magnesia | MgOH2 |
| Blue vitriol(neela thotha)copper sulphate | CuSO4 |
| Lime water | Calcium hydroxide |
| Gypsum | CuSO4.2H2O |
| Alum | K2SO4.Al2(SO4)3.24H2O |
| Slaked lime | Ca (OH) 2 |
| Caustic soda | NaOH |
| Caustic potash | KOH |
| Aqua Regia | 3HCl + HNO3 |
| Nausadar | NH4Cl |
Artificial Rain
- It refers to inducing precipitation by injecting certain chemicals into clouds to accelerate the process of raindrop formation.
- Cloud seeding involves dispersing substances like:
- Silver iodide (Agl)
- Potassium iodide
- Sodium chloride
- Dry ice (CO₂)
- These act as condensation nuclei, attracting water droplets, which then combine to form rain.
- Conditions Required: Potential rain-bearing clouds, sufficient humidity and atmospheric instability & Favorable wind and temperature conditions.
Rare Earth Elements (REEs)
- The Rare Earth Elements (REEs) are 17 metals in Group 3 of the Periodic Table comprising Lanthanide series elements and Scandium and Yttrium (due to similar physical properties and found in the same ores and deposits).
- Contrary to their name, they are moderately abundant in nature but not concentrated enough to make them economically exploitable. India has the fifth-largest rare earth resource globally, which are mainly concentrated in its monazite minerals.
- Rare Earth Elements are very important for futuristic technology.
- They have wide applications in clean energy, high-end electronics, defence, etc with no equal substitutes.

Instruments and the Physics Principle they work on.
1. Based on Archimedes’ Principle (Buoyancy & Density)
- Lactometer → Purity of milk (density).
- Hydrometer → Density of liquids.
- Ship/Submarine → Floating & sinking.
2. Based on Pascal’s Law (Pressure Transmission in Fluids)
- Hydraulic press / Brakes / Lift → Force multiplication in fluids.
- Syringe → Fluid pressure transmission.
3. Based on Boyle’s Law / Gas Laws
- Sphygmomanometer → Blood pressure measurement (gas pressure).
- Air pump / Bicycle pump → Pressure–volume relation.
4. Based on Bernoulli’s Principle
- Atomizer / Perfume sprayer
- Aeroplane wings (Lift)
- Carburetor in engines
5. Based on Electromagnetic Induction (Faraday’s Law)
- Transformer → Voltage step-up/step-down.
- Electric generator (dynamo) → Mechanical energy → Electrical energy.
- Induction stove → Heating by eddy currents.
6. Based on Thermodynamics / Heat
- Steam engine → Heat engine principle.
- Refrigerator & Air Conditioner → Second Law of Thermodynamics (heat pump).
- Thermocouple → Seebeck effect.
7. Based on Optics (Reflection / Refraction / Diffraction)
- Periscope → Reflection by plane mirrors.
- Telescope / Microscope → Refraction by lenses.
- Spectrometer → Refraction & diffraction.
- Prism → Dispersion of light.
- Camera → Refraction & image formation.
- Optical fibres → Total internal reflection.
8. Based on Osmosis / Dialysis
- Artificial kidney → Osmosis.
- Desalination plant → Reverse osmosis
9. Based on Radiation / Nuclear Physics
- X-ray tube → Sudden deceleration of electrons (Bremsstrahlung).
- Geiger–Müller counter → Ionization by radioactive particles.
- Cyclotron → Lorentz force & circular motion of charges.
|
Phenomenon |
Real life examples |
|
Refraction |
|
|
Scattering |
|
|
Reflection |
|
|
Reflection of sound |
|
|
Total internal reflection |
|
|
Dispersion |
|
